Molecular architecture
Molecular architecture. Chapter 2. Effects of chain composition and morphology on bulk properties. Outline. Macromolecule structure – building blocks, arrangement, interaction with the environment Synthetic polymers Carbon-carbon chains Carbon-nitrogen chains Carbon-oxygen chains

Molecular architecture
E N D
Presentation Transcript
Molecular architecture Chapter 2. Effects of chain composition and morphology on bulk properties chapter 2
Outline • Macromolecule structure – building blocks, arrangement, interaction with the environment • Synthetic polymers • Carbon-carbon chains • Carbon-nitrogen chains • Carbon-oxygen chains • Carbon-sulfur chains • Inorganic polymers • Copolymers • Types of constitutions and configurations • Conformations of single macromolecules • Structure/processing/performance chapter 2
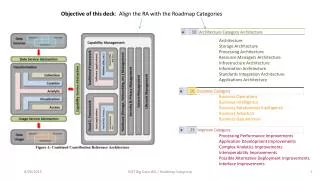
Macromolecular structure • Materials engineering is built on structure-processing-property relationships • Usually, three factors are used to define the 3-D structure (which leads to the bulk properties) • Constitution: the types of atoms in the chain, including side groups, end groups, and the molecular weight distribution • Configuration: the arrangement of these atoms in the chain and branch segments • Conformation: the 3-D arrangement of the chain in the media (solvent, polymer, …) • Constitution and configuration are established during synthesis, while conformation is affected by the media and its conditions chapter 2
Macromolecular structure -polyacrylics chapter 2
Polyacrylics - PMMA • Commercial monomers: methacrylic acid, methyl methacrylate, ethyl methacrylate, n-butyl methacrylate, isobutyl methacrylate, 2-hydroxyethyl methacrylate • Tg related to the size of the –OR group, with larger groups have higher Tg’s • Free radical polymerization is typical • Mw > 100,000: uniform properties • PMMA has very high light transmission over the visible range; alternative to glass • Compatible with human tissue – biomed. Appls. • Photoresist for ebeam lithography • Worldwide capacity ~ 1 B tons/yr chapter 2
Polyacrylics - PAA • An example of a commercial nonionic polymer soluble in water at room temperature • Water solubility is a results of the high number of polar and hydrogen-bonding groups per repeating unit • Free radical polymerization in water or suspension polymerization in benzene • Tg ~ 126 C; solid polymer is hard, clear and brittle • Viscosities of aqueous solutions increase with molecular weight • Dispersant for organic pigments, flocculant, adhesive, thickener • Worldwide capacity ~ 1 M tons/yr chapter 2
Polyacrylics- PAM • Polymerized via free radical initiators (azo compounds, redox catalysts, light, radiation) • Unique among vinyl and acrylic monomers because it will polymerize to very high molecular weights (~106 +) [high purity monomer, very high ratio of polymerization to termination rate constants] • Polymerizations: solution, inverse emulsion, inverse microemulsion, precipitation/suspension • Tg ~ 165 C • Slow dissolution in water, but suspensions are very stable • Flocculant [major market in water treatment applications], rheology control, adhesives • Worldwide monomer capacity ~ 1.4108 kg/yr chapter 2
Polyacrylics - PAN • Manufactured by the ammoxidation of propylene; route is low cost and led to high volume applications • Worldwide capacity ~4 B tons/yr • Head-to-tail configuration; isotactic and syndiotactic • Highly crystalline, hard, chemical resistance, low gas permeability, Tm ~ 317 C but the polymer decomposes below this temperature; solvents must be strongly hydrogen bonding with nonpolar segments • Excellent barrier properties to O2, CO2; monomer is highly toxic, • Acrylic fibers (50 % of monomer usage; easy to dye), copolymers with styrene (SAN) and butadiene + styrene (ABS) chapter 2
Polyacrylics – polyacroleinPoly(vinyl formaldehyde). Specialty polymer • Acrolein (2-propenal) is highly toxic and carcinogenic • Anionic, cationic catalyst lead to irregular structures • Free radical initiators lead to 1,2 vinyl addition, comb-like structures, which hydrolyze to ladder-like structures • A number of copolymers are possible The reaction product is insoluble; the tetrahydropyrane rings and possible interchain hemiacetal links contribute to its insolubility. S. Slomkowski, Prog. Polym. Sci., 23, 815-874 (1998). chapter 2
Polyacrylics – polyacroleinPoly(vinyl formaldehyde). Specialty polymer Conversion to the polyacrolein hydrate makes a water-soluble system suitable for medical applications S. Slomkowski, Prog. Polym. Sci., 23, 815-874 (1998). chapter 2
Polyacrylics – polyacroleinPoly(vinyl formaldehyde). Specialty polymer Core-shell particles with reactive surface groups can be carriers for covalently immobilized catalysts, enzymes, drugs and biopolymers S. Slomkowski, Prog. Polym. Sci., 23, 815-874 (1998). chapter 2
Polyacrylics – polyacroleinPoly(vinyl formaldehyde). Specialty polymer Example application. Diagnostic test to detect antibodies in blood. Direct. Antibodies are attached to the microspheres. When antigens are present, the microspheres agglomerate. Reverse. Antigens are immobilized to microspheres, and the presence of antibodies causes agglomeration. chapter 2
Polyacrylics – P(HEMA) • Synthetic bone • Interpenetrating polymer networks with collagen, polycaprolactone, vinyl pyrrolidone • Artificial corneas; drug delivery system • Hydrogels: swell in aqueous media. Dental cements, controlled drug release, prostheses, optical lenses chapter 2
G.D. Winter and B. J. Simpson, Heterotropic bone formed in a synthetic sponge in the skin of young pigs, Nature, 223, 88-90 (1969). Polyacrylics – P(HEMA) • Monolithic or heterogeneous (microporous or macroporous) 3-D crosslinked gels or sponges • Linear or branched polymers • Poly(HEMA) as bone regeneration medium; however, the synthetic polymer often is encapsulated, which precludes hard tissue replacement. • Poly(HEMA)/collagen composites: no fibrous tissue capsule, slow biodegradation, phosphatase activity in the implant fragments, uniform mineralization • Collagen influences adhesion, spreading, proliferation and differentiation of cells • The collagen distribution in the copolymer affects these results chapter 2
Polyacrylics – P(HEMA) Core(transparent p(HEMA))-and-skirt (opaque p(HEMA)) morphology Attachment of skirt to core via IPN (interpenetrating polymer network). Porous hydrogel allows cellular invasion, production of neocollagen, vascularization. chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
Green engineering – polymer examplesJ.L. Anthony, Kansas State University chapter 2
cArbon-carbon backbone chapter 2
Polydienes • Monomers either have two C=C bonds per unit (butadiene, isoprene, chloroprene) or one C=C in a ring (norbornene, pentenamer); stereochemistry controlled by polymerization conditions • Tg’s are lower than room temperature, crosslinking results in 3-D solids chapter 2
Polydienes - Tg chapter 2
Polyhalogens • The bond strengths of C-X are ordered: C-F > C-Cl > C-Br > C-I. The fluorocarbons are most stable • Teflon = PTFE; high thermal stability, resistant to corrosive attack • Kynar = PVDF; good weathering, architectural coatings • PVC; high volume thermoplastic, easy to compound • Saran = PVDC; flexible films chapter 2
Polyolefins • Low cost, high volume thermoplastics • PE; HDPE, LDPE,LLDPE, HMWPE • PP • PS chapter 2
Other vinyl polymers • Monomers: CH2=CHR • PVAc: poly(vinyl acetate). Adhesives, glues, emulsions, suspensions • PVAL: poly(vinyl alcohol). Water-soluble • Poly(vinyl ethers). Adhesives and plasticizers chapter 2
Aromatic w/C-C backbone • Poly(p-xylylenes). Specialty coatings • Phenolic resins. • Novolacs via acid catalysis. Soluble in alcohols • Bakelites via base catalysts. Crosslink with heat. Early application – distributor cap. • Pine oils. b-pinene chapter 2
Carbon-nitrogen backbone chapter 2
Polyamides • [-NH-CO-] in the main chain • Perlon – repeating unit and the monomer are the same. “Nylon X”. [-NH-CO-R-]. Example – Nylon 6. poly(e-caprolactam). Ring-opening lactam polymerizations may have significant monomer levels. • Nylon – repeating unit is formed with two monomers. “Nylon X,Y” [-NH-R-NH-CO-R1-CO-]. Example – Nylon 6,6. poly(hexamethylene adipamide). Equilibrium is far to products. • Nomex, Kevlar have aromatic “R” groups chapter 2
Polyimides • [-NH-CO-] • Kapton – very rigid chain • Nylon 1 – poly(isocyanic acid) chapter 2
Polyurethanes Typical monomer pairs are diols/diisocyanates. Polymers have wide ranges of stiffness, hardness and densities due to the wide variety of monomers available; low density flexible foams, soft solid elastomers and print rollers, and high performance adhesives. Original materials were developed by Otto Bayer, 1937, I.G. Farben. The monomers circumvented the patents by W. Carothers (du Pont) for polyesters. A major commercial product was polyurethane foam based on toluene diisocyanate and polyester precursors. Foaming occurred due to the heating of adventitious water in the reaction mixture. chapter 2
Polyurethanes - history • Otto Bayer, 1937, I.G. Farben. Liquid diisocyanates and liquid polyether or polyester diols (circumventing patents by Carothers (du Pont) on polyesters) • TDI + polyester polyols, 1954, flexible foams due to adventitious water • Linear fibers from hexamethylene diisocyanate (HDI) and 1,4-butanediol • Poly(tetramethylene glycol), duPont, 1956. 1st polyether diol. Low cost, ease of handling causes replacement of polyesters. BASF, Dow have products • 1960’s: chlorofluoroalkane blowing agents, cheap polyether diols, methylene diphenyl diisocyanate (MDI) for rigid foams, polyisocyanurates for better thermal stability. Auto interior safety components – instrument panels with semi-rigid foams chapter 2
Polyurethanes - history • Bayer AG introduces RIM (reaction injection molding), 1969, all plastic car • Polyurethane RIM – diamine chain extenders, trimerization; poly(urethane urea), poly(urethane isocyanurate), polyurea RIM • Fillers ( mica, milled glass, mineral fibers) – RRIM, for improved flex modulus for stiffness and thermal stability. Leads to the 1st plastic body auto in US (Pontiac Fiero, 1983). SRIM – structural RIM with glass mats • 1980’s – auto market share increased via PVC plastisol replacement • 1990’s – Montreal Protocol leads to HCFC’s replacing CFC’s, and then CO2, pentane, 1,1,1,2-tetrafluoroethane (HFC-134a) and 1,1,1,3,3-pentafluoropropane (HFC-245fa) • 2-component polyurea spray elastomers for coatings on concrete and steel, 2-component polyurethane and hybrid polyurethane-polyurea elastomers for spray-in-place bed liners • 2004 – polyols derived from vegetable oils chapter 2
Polyurethanes - chemistry • Reaction polymers – polyurethanes, epoxies, unsatruated polyesters, phenolics • Polyaddition of polyisocyanate with a polyol • R1 – N=C=O + R2-O-H R1-NH-CO-O-R2 • Isocyanates react with any active hydrogen; with water, form a urea linkage and CO2 • Commercial products – liquid isocyanate, polyols, catalyst, additives (chain extenders, crosslinkers, surfactants, flame retardants, blowing agents, pigments, fillers) • Polymeric MDI (diphenylmethane diisocyanate) – blend of 2, 3, and 4 functional monomers with 2.7 average functionality for crosslinking • Prepolymer – partially reacted isocyante + polyol • Key isocyanate properties – backbone, % NCO, functionality, viscosity • Polyols – polyethers (PO, EO) or polyesters (adipic acid + EG)- backbone, initiator, MW % primary hydoxyls, functionality viscosity chapter 2
Polyurethanes - chemistry • Polymerization catalyzed by tertiary amines (dimethyl cyclohexylamine) and organometallic salts (dibutyl tin laurate) • Gel reaction (urethane), blow reaction (urea), trimerization reaction (potassium octoate) • Blowing agents – BP near room temperature, heat of reaction provides energy to vaporize the agent, bubbles grow at nucleation sites, polymer must cure prior to bubble aggregation and growth, surfactants can help control whether closed cell foams or open cell foams are made chapter 2
Polyurethanes - toxicity • CAS # 9009-54-5: polyurethane is chemically inert, no exposure limits by OSHA, ACGIH; not regulated for carcinogenicity • Combustible solid, decomposing to CO, NOx, hydrogen cyanide. • Dust may irritate the eyes and lungs • Isocyanates: skin and repiratory sensitizers • Polyols: may contain regulated compounds • Info: Polyurethane Manufacturers Association, Center for the Polyurethane Industry, Code of Federal Regulations Title 21 (Food and Drugs) and Title 40 (Protection of the Environment) chapter 2
Polyurethane – material propertieshttp://en.wikipedia.org/wiki/Polyurethanes chapter 2
Polyurethanes – structure, processing, properties • Diisocyanate, polyol, catalyst, surfactants • Density: vary the type of monomer • Cure rate: functional group reactivity, # of functional isocyanate groups • Photostability: type of diisocyanate, aromatics yellow with exposure to light • Soft, elastic, good flex: linear difunctional PEG segments (Spandex, soft rubber parts, foam rubber) • Rigid products: polyfunctional polyols for 3-D crosslinked structure, trimerization catalysts create cyclic structures high thermal stability • Memory foam: control of viscoelastic properties via catalysts and polyols to make the product softer at room temperature chapter 2
Polyurethanes - applications • Varnish: finish coats, hardwood floor sealing, faster and higher film build, cure occurs after solvent evaporation and with moisture; exterior varnishes susceptible to UV damage • Solid elastomer wheels: roller blading and skateboarding, abrasion resistance • Furniture: batch processes to make foam cushions, casting soft edges for table tops and panels, bottoms of mouse pads • Vehicle seats: seats, headrests, armrests, roof liners, instrument panels; in-situ foam-in-fabric (direct molding – seat cover on mold surface, metal framework insert, inject two-part mixture through mixing head under vacuum) • Decorative elements for houses: domed ceilings, statues, … chapter 2
Polyurethanes - applications • Watercraft: core foam for surfboards, boat hulls • Construction sealants: 1,2 and 3 part systems for rapid cure sealants • Tennis grips: Yonex Supergrap™, • Electronic components: “potting” or enclosure material to protect circuit boards, 121 C is upper temperature limit; two-part cast urethane • Adhesives: Gorilla Glue™, excellent water resistance; book binding adhesive; preferable to hotmelt and cold glue, 0.03 mm thickness, -40 C to 100 C chapter 2
CARBON-Oxygen backbone chapter 2
Polyacetals • [-CHR-O-] • Polymerization of aldehydes or cyclic trimers. Example – poly(oxymethylene) from formaldehyde or trioxane • Engineering thermoplastics, good abrasion and wear characteristics chapter 2
Polyethers • [-R-O-] • Aliphatic: PEO, water soluble; PPO – polyurethane intermediates, lubricants, surfactants; EO/PO copolymer – water soluble detergents and elastomers; Poly(tetrahydrofuran) – thermoplastic elastomers; poly(epichlorohydrin) - coatings chapter 2
Polyethers • Aromatic: epoxies based on bisphenol A; poly(phenylene oxide); PEEK – poly(ether ether ketone) • PEEK: E = 3.6 GPa, tensile = 90 MPa, 50% elongation at break • Tg’s at ~140 C and ~275 C based on composition; Tm ~ 350 C • Bearings, piston parts, pumps, UHV applications • Reinforced with carbon fiber for medical implants, aerospace structural material • Electronic circuitry (high temperature) • Resistant to many chemicals and solvents chapter 2
Polyesters - aliphatic • Ester group in the chain + aliphatic group in repeating unit • Step polymerization: ring-opening, self-condensation, transesterifications, diols + dicarboxylic acids or diacyl chlorides, acid anhydrides + cyclic ethers, o-carboxylic anhydrides • Copolymers of lactic and glycolic acids make biodegradable polymers for tissue engineering, drug delivery, and other biomedical uses chapter 2
Polyesters – PLA, PGA • Bacterial fermentation of corn starch to lactic acid • Dimer is catalytically (stannous octoate) polymerized • lactic acid is chiral; poly(L-lactic acid) and poly(D-lactic acid) are crystalline; PLLA - Tm ~ 60 C, Tm ~ 173 C • tissue engineering scaffolds: • Molecular self-assembly • Non-woven technology – PGA • Solvent casting and particulate leaching - PLA in dichloromethane + inorganic salts; film casting, leaching of the salt • Gas foaming • Emulsification/freeze drying • Liquid-liquid phase separation chapter 2
Polyesters - crosslinked • Ester monomers with C=C in the chain allow polyester polymerization + free radical crosslinking, leading to 3-D polymers • Crosslinked polyesters are the continuous phase for fiberglass polymers – boat hulls, vehicle bodies, etc. – as sheet molding compound • These systems are usually manufactured in several steps: 1) viscous prepolymer is mixed with the glass fibers to make a sheet, 2) the sheet is placed in a mold and heated, increasing the polymerization of the polyester and permitting free radical polymerization of the C=Cs chapter 2