Le Chatelier’s Principle
831 likes | 5.26k Views
Le Chatelier’s Principle. When a system is in equilibrium and a stress is added to the system, the system will shift in order to alleviate itself of the stress in order to return to equilibrium Stresses: 1. concentration changes 2. temperature changes 3. pressure changes.

Le Chatelier’s Principle
E N D
Presentation Transcript
Le Chatelier’s Principle • When a system is in equilibrium and a stress is added to the system, the system will shift in order to alleviate itself of the stress in order to return to equilibrium • Stresses: 1. concentration changes 2. temperature changes 3. pressure changes
Concentration Changes • Adding a substance shifts the equilibrium away from the added substance in order to use the excess amount up and to reestablish equilibrium A + B C Add A, shift right (away from A) Add B, shift right (away from B) Add C, shift left (away from C) • Removing a substance shifts the equilibrium toward the substance in order to replenish the amount removed and to reestablish equilibrium A + B C Remove A, shift left (toward A) Remove B, shift left (toward B) Remove C, shift right (toward C)
Temperature Changes • Based upon whether the reaction is endothermic (takes in heat) or exothermic (gives off heat) • Follow the same rules as concentration – • Add heat, shift away from the side heat is located on • Remove heat, shift toward the side heat is located on A + B C + heat Add heat, shift left (away) Remove heat, shift right (toward) A + B + heat C Add heat, shift right (away) Remove heat, shift left (toward)
Pressure Changes • ONLY affects GASES • Use coefficients to determine the total # of gases of products and reactants • Increase pressure, shift towards to the side with the fewest moles of gas • Decrease pressure, shift towards the side with the greatest moles of gas A(g) + B(g) C (g) 1 mole product gas vs. 2 moles of reactant gas Increase pressure, shift right (fewest moles) Decrease pressure, shift left (greatest moles)
Additional Stuff • Adding a catalyst has no affect on the equilibrium system • Adding an inert gas (gas that doesn’t react) or a gas not in the equilibrium system, has no affect on the equilibrium • The only change that will affect the value of K is a temperature change.
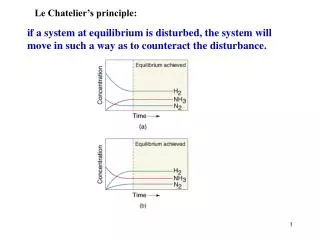
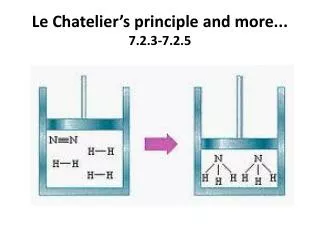
Example N2(g) + 3H2(g) 2NH3(g) + 92 kJ/mol What affect does each of the following have on the equilibrium system? What direction will the equation shift? • Adding H2shift right • Removing NH3shift right • Decreasing volume V, P, shift right (fewest mols) • Decreasing pressure shift left (greater mols) • Increasing temperature shift left • Adding a catalyst no affect
















![Definition pH and pOH. Given pH, pOH, [H 3 O + ] or [OH¯], calculate the remaining values.](https://cdn2.slideserve.com/5054819/slide1-dt.jpg)
