Functionalized Composite Electrodes for Electrocatalytic Hydrogenation
11 likes | 178 Views
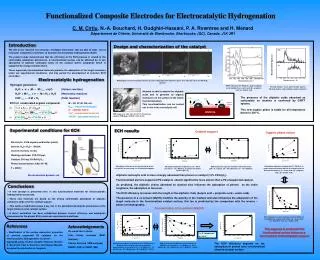
2. 4. 3. 1. Hydrogen generation: H 3 O + + e - + M ↔ M H ads + H 2 O (Volmer reaction) H 3 O + + M H ads + e - ↔ M + H 2 + H 2 O (Heyrovsky reaction) 2 M H ads ↔ 2 M + H 2 (Tafel reaction). 10% Pd/Al 2 O 3. A l. Pd. O.

Functionalized Composite Electrodes for Electrocatalytic Hydrogenation
E N D
Presentation Transcript
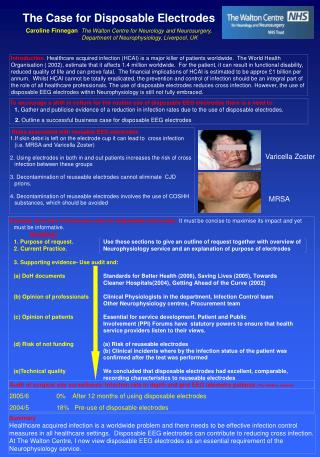
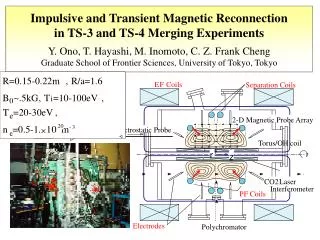
2 4 3 1 Hydrogen generation: H3O+ + e- + M ↔MHads + H2O (Volmer reaction) H3O+ + MHads + e-↔ M + H2 + H2O (Heyrovsky reaction) 2 MHads↔ 2 M + H2(Tafel reaction) 10% Pd/Al2O3 Al Pd O DRIFT spectra of the Pd/Al2O3 catalyst aliphatic acids modified: (a) - acetic acid; (b) - propionic acid; (c) - butyric acid Thermal analysis - mass spectroscopic data for Pd/Al2O3 butyric acid modified catalyst (under Ar) ECH of unsaturated organic compound: Y=Z + A↔ (Y=Z)adsA (Y=Z)adsA + 2MHads↔ (YH-ZH)adsA (YH-ZH)adsA↔YH-ZH + A M – Pd, Pt, Ni, Rh, etc. Hads – adsorbed hydrogen A – catalyst matrix Y=Z – unsaturated molecule YH-ZH – saturated molecule TEM image of a ultra thin cut of 10% Pd-alumina catalyst (TEM Mag = 200000 x; HV= 80 kV) Experimental conditions for ECH Electrolyte: 0.5 M organic acid buffer (pH=5); Solvent: H2O / H2O – MeOH; Current intensity: 20 mA Working electrode: CVR 100 ppi; Catalyst: 200 mg 10%Pd/Al2O3; Phenol concentration: 8.8510-3 M; T = 298 K; Adsorption isotherms of (▲)phenol and (■) acetic acid (pH=5) in waterusing 10% Pd-alumina supports. Adsorption isotherms of phenol on 10% Pd/Al2O3 in water: methanol solution (80:20v/v) in the presence of different electrolytes: (●) - acetic acid; (■) - propionic acid; (▲) - butyric acid; ECH of phenol in aqueous medium 0.5 M acetic buffer (pH = 5); catalyst: (■) – Pd submicron; (●) – 10% Pd/Al2O3; Adsorption isotherms of phenol on: (■) – Pd 63 μm and (●) – 10% Pd/Al2O3 in 0.5 M acetic buffer solution (pH = 5); Electrochemical dynamic cell ECH of phenol in water: methanol solution (80:20v/v) using different support electrolyte: (■) - acetic acid; (●) - propionic acid; (▲) - butyric acid; Adsorption isotherms of phenol on 10% Pd/Al2O3 in 0.5 M acetic buffer solution (pH = 5) for different concentrations of co-solvent: (♦) – 0 % MeOH; (●) – 5 % MeOH; (■) – 20 % MeOH; (▲) – 50 % MeOH; T = 323 K; ECH of phenol using different concentrations of MeOH as co-solvent: (♦) - 0 % MeOH; (●) - 20 % MeOH; (■) - 50 % MeOH; (▲) - 60 % MeOH; T = 298 K; Functionalized Composite Electrodes for Electrocatalytic Hydrogenation C. M. Cirtiu, N.-A. Bouchard, H.Oudghiri-Hassani, P. A. Rowntree and H. Ménard Département de Chimie, Université de Sherbrooke, Sherbrooke, (QC), Canada, J1K 2R1 Introduction Design and characterization of the catalyst The aim of our research is to develop «intelligent electrodes» that are able to make use of molecular recognition at interface to facilitate electrocatalytic hydrogenation (ECH). The present study demonstrates that the efficiency of the ECH process is related to the controllable adsorption phenomena. A functionalized surface can be obtained by in situ adsorption of aliphatic carboxylic acids on the catalyst matrix, adsorption which is supported by energy considerations. These organically functionalized materials promote the adsorption of the target molecules under our experimental conditions, and may permit the development of selective ECH electrodes. Electrocatalytic hydrogenation Micrographics of the secondary electrons (1) and cartography of the elements (2,3,4) for a ultra thin cut of 10% Pd/Al2O3 catalysts Alumina is able to adsorb the aliphatic acids and to generate an organic monolayer on the surface of the matrix (functionalization). This functionalization can be carried out in situ in the electrolysis cell. The presence of the aliphatic acids adsorbed as carboxylate onalumina is confirmed by DRIFT spectra. This new organic phase is stable for all temperature below to 200 ºC. ECH results Catalyst support Organic phase nature Aliphatic carboxylic acid is more strongly adsorbed than phenol on catalyst (10% Pd/Al2O3). Functionalized alumina supported Pd catalysts adsorb significantly more phenol than a Pd unsupported catalyst. As predicted, the aliphatic chains adsorbed on alumina also influence the adsorption of phenol; as the chain lengthens, the adsorption is favoured. The ECH efficiency increases with the length of the aliphatic chain (butyric acid > propionic acid > acetic acid). The presence of a co-solvent (MeOH) modifies the polarity of the medium and also influences the adsorption of the target molecule to the functionalized catalyst surface; this too is predicted by the comparison with the reverse –phase chromatography. Conclusions • A new concept is presented here:in situ functionalized materials for electrocatalytic hydrogenation processes. • These new materials are based on the strong controllable adsorption of aliphatic carboxylic acids onto the catalyst support. • This surface modification plays a key role in the adsorption/desorption phenomena of the target molecule onto catalyst surface. • A direct correlation has been established between current efficiency and adsorption phenomena for the phenol ECH, under our experimental conditions. Concentration of co-solvent (MeOH) References « Modification of the surface adsorption properties of alumina supported Pd catalysts for the electrocatalytic hydrogenation of phenol» Ciprian M. Cirtiu, Hicham Oudghiri Hassani, Nicolas-A. Bouchard, Paul A. Rowntree and Hugues Ménard, accepted for publication in Langmuir Acknowledgements We would like to thank: Irène Kelsey Lévesque (SEM analyses) Charles Bertrand (TEM analyses) NSERC ($$$) & FQRNT ($$$) This sequence is predicted if the functionalized surface behave as a reverse-phase chromatographic support Ce= 0.1 µmole mL-1 Q= 100 C The ECH efficiency depends on the adsorption of phenol onto functionalized alumina catalyst surface