Understanding Ozone Depletion: Causes, Effects, and Solutions
Ozone depletion is a critical environmental issue that results from the thinning of the ozone layer, primarily caused by human-made chemicals like chlorofluorocarbons (CFCs). The ozone layer acts as Earth's sunscreen, shielding life from harmful ultraviolet (UV) radiation. Increased exposure to UV radiation can lead to serious health issues, including skin cancer and eye damage, as well as detrimental effects on marine ecosystems and agriculture. Awareness and action are key to reversing this trend and protecting our planet.

Understanding Ozone Depletion: Causes, Effects, and Solutions
E N D
Presentation Transcript
By: Tori, Alyssa & Jessica Ozone Depletion
What is Ozone? • Ozone is a gas that is made up of three oxygen atoms (03). Ozone occurs naturally in small amounts in the upper part of the atmosphere (the stratosphere). Ozone protects life on earth from the suns ultraviolet UV radiation (its been called the earths sunscreen).
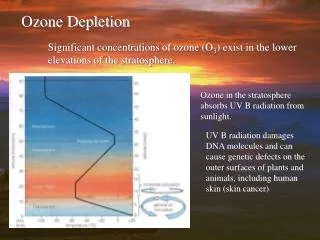
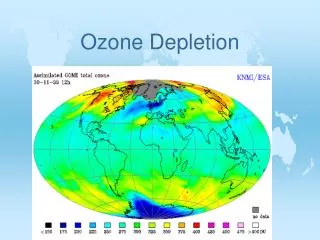
Ozone Depletion • In the 1980's, scientists found evidence that the ozone layer was being depleted. • Depletion of the ozone layer results in increased UV radiation reaching the earths surface, which in turn leads to a greater chance of over exposure to UV radiation. • Ninety percent of the ozone in the atmosphere sits in the stratosphere. The natural level of ozone in the stratosphere is a result of a balance between sunlight that creates ozone and chemical reactions that destroy it. • Ozone is created when the kind of oxygen we breathe,O2, is split apart by sunlight into single oxygen atoms. Single oxygen atoms can re-join to make O2, or they can join with O2 molecules to make ozone (O3)
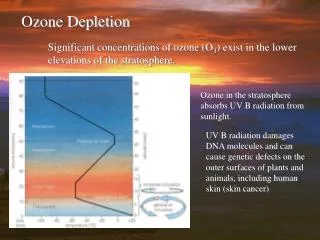
Ozone is destroyed when it reacts with molecules containing nitrogen, hydrogen, chlorine, or bromine. Some of the molecules that destroy ozone occur naturally, but people have created others. • Ozone in the stratosphere absorbs most of the ultraviolet radiation from the Sun. Without ozone, the Sun’s intense UV radiation would sterilize the Earth’s surface. Ozone screens all of the most UV-c radiation and most of the UV-b radiation. Ozone only screens about half of the UV-a radiation. Excessive UV-b and UV-a radiation can cause sunburn and can lead to skin cancer and eye damage.
Solar ultraviolet radiation is largely absorbed by the ozone in the atmosphere—especially the harmful, high-energy UV-a and UV-b. The graph shows the flux (amount of energy flowing through an area) of solar ultraviolet radiation at the top of the atmosphere (top line) and at the Earth’s surface (lower line). The flux is shown on a logarithmic scale, so each tick mark on the y-axis indicates 10 times more energy. Without the ozone protecting us from these rays, it would be very difficult for anything on earth to survive.
Chemicals that break down ozone • One of the leading cause of ozone depletion is the use and emission of CFC's (chlorofluorocarbons) • CFC's are a man made chemical that was produced as a safer alternative to refrigerants • This chemical consist of a chlorine atom, a fluorine atom, and a carbon atom • These are non toxic chemicals that are non-reactive and are very stable near the earth's surface • Discovered in 1930's by Thomas Midgley, an American chemist After a while, people began to realize that once this chemical enters the stratosphere, the chlorine atom is removed and attracts one of the three oxygen atoms found in the ozone molecule • This is a continuous process and over 100,000 ozone molecules can be destroyed by a single chlorine atom • A study was done in 1974 by Sherwood Rowland and Mario Molina. They found proof through research that CFC's were entering the atmosphere, and found that 99% of CFC molecules would enter the stratosphere • CFC’s were very cheap and effective, so many people argued against the harmful accusations of CFC’s • In 1984, the ozone hole was discovered over Antarctica and there was no more arguments of the destructive causes of the CFC's
Sources of these chemicals include refrigerators, home insulation, aerosol spray propellants, plastic foam, old air conditioners and food containers.
Environmental Effects • Large effect on marine life • It was found that solar UVB radiation (a very harmful ultraviolet light) causes extensive damage to the early stages of life in fish, shrimp, crab, etc • It causes a reduction in reproduction capabilities, and impairs larval development (when the embryo develops into a larva instead of directly into a organism) • Ozone depletion will cause a disruption in life cycles; therefore, causing a change in the food chain which will have a negative effect on many animals • Oceans will also be largely effected resulting in a decreased survival rate of basic organisms such as plankton • This will cause many animals above plankton on the food change to die out and results in a lowered biodiversity • Wheat, rice, barley, oats, corn, soybeans, peas, tomatoes, cucumbers, cauliflower, broccoli and carrots will all be effected as they are all extremely sensitive to UV rays which will result in a reduction of growth, photosynthesis, and flowering
Social Effects • Whenever 1% of the ozone is depleted, 2% more of UVB is able to reach the Earth • As the UVB exposure increases, so will skin cancer levels • It is estimated by the EPA (Environmental Protection Agency) that 60 million Americans born by the year 2075 will get skin cancer as a cause of ozone depletion, and of these people, one million will die • The EPA also estimates that rates of malaria and other diseases will increase, and 17 million more cases of cataracts will be expected • A study of ozone depletion was done by the National Academy of Science of the United States where they described the effects of ozone depletion as: • Worst case, 70% of the ozone will decrease, causing a blistering sunburn after 10 minutes of exposure • More probable case, 50% of the ozone will deplete causing a blistering sunburn after 1 hour • Either way, this concludes that outdoor work in the northern hemisphere would require complete covering by protective clothing
Economical Effects • significant increase in replacing/protecting materials • There will be a massive impact on industries such as meat, fishing, agricultural, etc since the food chains are going to be heavily impacted, which will cause the endangerment or extinction of some animals • Death rates will also increase as a result of the rise of skin cancer • Third world countries rely very heavily on working during the daylight hours • With the increase in UVB, it causes an inability for workers and cattle to work outdoors, which can cause massive reductions in crop production and consequently leads to famine
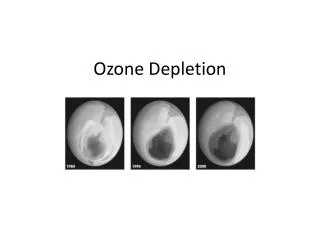
Human Health Affects • Skin Cancer – More new cases of skin cancer are diagnosed than new cases of breast, prostate, lung and colon cancer. Unprotected exposure to UV radiation is the most preventable risk factor for skin cancer. • Melanoma – Melanoma is the most serious form of skin cancer. This causes more than 75 percent of skin cancer deaths. This is due to UV exposure and sunburn diseases. • Nonmelanoma Skin Cancers – These skin cancers are less deadly than melanoma. If they are left untreated they may spread. Two common types of nonmelanoma skin cancers are: basal cell and squamous cell carcinomas. Basal cell is the most common type of skin cancer. They usually appear as small, fleshy bumps on other skin areas and rarely spread to other parts of the body. Squamous cell carcinomas tumors may appear as nodules or as red, scaly patches. This cancer can spread to other parts of the body. • Chronic exposures to the sun also causes premature aging, which over time can make the skin become thick, wrinkled and your skin ages itself many years. • Cataracts and Other Eye Damage– Cataracts are a form of eye damage in which a loss of transparency in the lens of the eye clouds vision. If it is left untreated, cataracts can lead to blindness. Other kinds of eye damage include pterygium which is tissue growth that can block vision. Also skin cancer around the eyes, and degeneration of the macula (the part of the retina where visual perceptions is the most)
Montreal Protocol • The Montreal Protocol establishes legal controls on the national production and consumption of the ODS (ozone depleting system). • Today 191 countries have signed to this treaty. These countries are committed to taking action to reduce and eliminate ODS to protect the ozone layer. • The Montreal Protocol has been successful because of scientific accuracy, effective public policy, and market based, flexible, innovated approaches to ensuring the ozone layer protection. • If all countries that have signed the Protocol maintain their commitments, the decrease of ODS in the atmosphere will continue through the 21st century.
Solutions • Some solutions to the problem are the Montreal Protocol. • This is the biggest solution in place right now for the depletion of the ozone layer. • Also, the National Resources Defense Council asked the CFC makers to only eliminate half of their CFC production • In the year 2000, the US and twelve nations in Europe have agreed to ban all use and production of CFCs. • Also possible solutions are to minimize high altitude aircraft flights (oxygen reduction and water vapor deposition), encourage growth of plants that produce oxygen, and to discourage deforestation. • Another solution is to immediately stop producing ozone depleting chemicals. • Even with immediate action, models indicate that it will take 50-60 years for the ozone layer to return in 1975 levels and another 100-200 years for full recovery to pre 1950 levels.
Perspectives • Many people believe that ozone depletion will result in many health complications. • Sherry Rowland and Mario Molina, chemists at the University of California, published the first scientific paper warning that human-generated chlorofluorocarbons (CFCs) can cause serious harm to the Earth's protective ozone layer. They warned that the loss of ozone would significantly increase the amount of skin-damaging ultraviolet UV-B light reaching the surface which would greatly increase skin cancer and cataracts. • The loss of ozone could also significantly cool the stratosphere, potentially causing destructive climate change. Although no stratospheric ozone loss had been observed yet, CFCs should be banned.
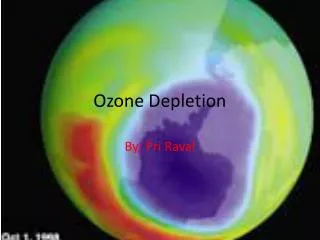
Antarctica is the first known case of ozone depletion. Scientists found a large hole in the ozone right above this continent. Antarctica is now threatened by the many melting icecaps, due to the increase in UV radiation. • All scientists agree that in the future, ozone depletion will have to be solved, so that the protective ozone layer can be conserved. • Some scientists have a negative perspective when discussing the ozone, however there are others who focus on the positive. Now, researchers in Tasmania have found that the hole in the ozone is shrinking for the first time since the hole was discovered in 1985. Many of these scientists believe that we have already started correcting the problem, and that the ozone will continue to rebuild and fix itself.
Thank- You http://www.youtube.com/watch?v=qUfVMogIdr8