What is a volcano?
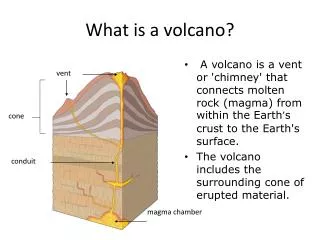
What is a volcano?. A hill with a crater? Does magma need to be involved? Does it matter?.

What is a volcano?
E N D
Presentation Transcript
What is a volcano? A hill with a crater? Does magma need to be involved? Does it matter? Lecture material about Introduction to Volcanology, covering, Heat in the earth, where magma comes from and how, earth’s mantle, tectonics and convection, basalt and why it is fundamental, where volcanoes are. Thanks to Wendy Bohrson and Glen Mattioli who provided many of the slides.
Magma Plumbing System • Melts form in mantle • Pool in magma chambers • Magma eventually erupts
Volcanology Study of generation of magma, transport of magma, and shallow-level or surface processes that result from intrusion and eruption of magma
Volcanology • Physical and chemical behavior of magmas • Transport and eruption of magma • Formation of volcanic deposits
What do we need for volcanism? • Thermal energy • Material to melt • Ability to erupt
Earth’s Energy Budget • Solar radiation: 50,000 times greater than all other energy sources; primarily affects the atmosphere and oceans, but can cause changes in the solid earth through momentum transfer from the outer fluid envelope to the interior • Radioactive decay:238U, 235U, 232Th, 40K, and 87Rb all have t1/2 that >109 years and thus continue to produce significant heat in the interior; this may equal 50 to 100% of the total heat production for the Earth. Extinct short-lived radioactive elements such as 26Al were important during the very early Earth. • Tidal Heating: Earth-Sun-Moon interaction; much smaller than radioactive decay • Primordial Heat: Also known as accretionary heat; conversion of kinetic energy of accumulating planetismals to heat. • Core Formation: Initial heating from short-lived radioisotopes and accretionary heat caused widespread interior melting (Magma Ocean) and additional heat was released when Fe sank toward the center and formed the core
What are the sources of heat within Earth? • Primordial/accretional energy • Radioactive decay
“Natural” Radioactivity • Elements (determined by Z) typically exist as a mix of isotopes which have different atomic weights (eg 39K and 40K, where Z=19). • Isotopes may be stable, radioactive or radiogenic. • 39K is stable, 40K is radioactive, 40A and 40Ca radiogenic. • Decay of radioactive isotopes has a very predictable rate: N = Noe-t . • This decay occurs spontaneously everywhere and is not influenced by changes in T, P or composition! • Decay reactions of many types occur: 40K-> 40Ca + electron + heat. • Discovered by Marie Curie.
Natural Radioactivity is exploited by volcanologists and petrologists. Radiometric dating. System of 40K->40A leads to K/A and A/A dating methodology. These use the age eqn and depend on purging of A at time of eruption. Radioactive Tracing. Use isotopic ratios of elements to tell where the magma came from. Ex: 87Sr/86Sr this is radiogenic/stable, so it can measure the amounts of radioactive parent= 87Rb
dN dN - µ - l N or = N dt dt Radioactive Decay The Law of Radioactive Decay # parent atoms time
D = Nelt - N= N(elt -1) age of a sample (t) if we know: D the amount of the daughter nuclide produced N the amount of the original parent nuclide remaining l the decay constant for the system in question
The K-Ar System 40K either 40Ca or 40Ar • 40Ca is common. Cannot distinguish radiogenic 40Ca from non-radiogenic 40Ca • 40Ar is an inert gas which can be trapped in many solid phases as it forms in them
l æ ö e ç ÷ è ø l The appropriate decay equation is: 40Ar = 40Aro + 40K(e-lt -1) Where le = 0.581 x 10-10 a-1 (electron capture) and l = 5.543 x 10-10 a-1 (whole process)
Blocking temperatures for various minerals differ • 40Ar-39Ar technique grew from this discovery
How do we know the composition of the mantle? • Peridotite bodies (e.g., ophiolites) • Xenoliths • Cosmochemical Evidence/Meteorites
Ophiolites Seismic velocity is plotted on the horizontal axis versus depth below the seafloor on the vertical axis. The different seismic layers are marked on the plot with geologic interpretations of the rock units. The layers are defined by velocities and velocity gradients. Cross section through a typical ophiolite sequence is shown to the right. http://www.womenoceanographers.org/doc/KGillis/Lesson/gillis_lesson.htm
Ophiolites Picture of a hillside in Cyprus. The vertical slabs of rock are dikes intruding into lavas that erupted on the seafloor. This section represents the transition from lavas to sheeted dikes and is thought to correspond to seismic Layer 2B as seen in Figure 5. Taken from the RIDGE field school in Cyprus. http://www.womenoceanographers.org/doc/KGillis/Lesson/gillis_lesson.htm
Mantle Xenoliths http://www.nhm.ac.uk/mineralogy/petrology/MantleXenoliths.htm
Carbonaceous Chondrites Left to right: fragments of the Allende, Yukon, and Murchison meteorites http://www.daviddarling.info/encyclopedia/C/carbchon.html
Composition of the Mantle What is the mineralogy of the mantle? Olivine +clinopyroxene + orthopyroxene ± plagioclase, garnet, spinel (Al bearing minerals)
crust obvious from space that Earth has two fundamentally different physiographic features: oceans (71%) and continents (29%) from: http://www.personal.umich.edu/~vdpluijm/gs205.html global topography
Differentiation of the Earth • Melts extracted from the mantle rise to the crust, carrying with them their “enrichment” in incompatible elements • Continental crust becomes “incompatible element enriched” • Mantle becomes “incompatible element depleted” From: http://www.geo.cornell.edu/geology/classes/geo302
Radioactivity in earth materials Heat production decreases with depth from crust to mantle.
Earth’s Geothermal Gradient Average Heat Flux is 0.09 watt/meter2 Geothermal gradient = DT/ Dz 20-30°C/km in orogenic belts; Cannot remain constant w/depth At 200 km would be 4000°C ~7°C/km in trenches Viscosity, which measures resistance to flow, of mantle rocks is 1018 times tar at 24°C ! Approximate Pressure (GPa=10 kbar)
Earth Interior Pressures • P = rVg/A = rgz, if we integrate from the surface to some • depth z and take positive downward we get • DP/Dz = rg Rock densities range from 2.7 (crust) to 3.3 g/cm3 (mantle) 270 bar/km for the crust and 330 bar/km for the mantle At the base of the crust, say at 30 km depth, the lithostatic pressure would be 8100 bars = 8.1 kbar = 0.81 GPa
Gravity, Pressure, and the Geobaric Gradient • Geobaric gradient defined similarly to geothermal gradient: DP/Dz; in the interior this is related to the overburden of the overlying rocks and is referred to as lithostatic pressure gradient. • SI unit of pressure is the pascal, Pa and 1 bar (~1 atmosphere) = 105 Pa • Pressure = Force / Area and Force = mass * acceleration • P = F/A = (m*g)/A and r (density) =mass/volume
Heat Flow on Earth An increment of heat, Dq, transferred into a body produces a Proportional incremental rise in temperature, DT, given by Dq = Cp * DT where Cp is called the molar heat capacity of J/mol-degree at constant pressure; similar to specific heat, which is based on mass (J/g-degree). 1 calorie = 4.184 J and is equivalent to the energy necessary to raise 1 gram of of water 1 degree centigrade. Specific heat of water is 1 cal/g°C, where rocks are ~0.3 cal/g°C.
Heat Transfer Mechanisms • Radiation: involves emission of EM energy from the surface of hot body into the transparent cooler surroundings. Not important in cool rocks, but increasingly important at T’s >1200°C • Advection: involves flow of a liquid through openings in a rock whose T is different from the fluid (mass flux). Important near Earth’s surface due to fractured nature of crust. • Conduction: transfer of kinetic energy by atomic vibration. Cannot occur in a vacuum. For a given volume, heat is conducted away faster if the enclosing surface area is larger. • Convection: movement of material having contrasting T’s from one place to another. T differences give rise to density differences. In a gravitational field, higher density (generally colder) materials sink.
Magmatic Examples of Heat Transfer Thermal Gradient =DT between adjacent hotter and cooler masses Heat Flux = rate at which heat is conducted over time from a unit surface area Thermal Conductivity = K; rocks have very low values and thus deep heat has been retained! Heat Flux = Thermal Conductivity * DT
Types of Thermal Energy Transfer Models of Earth’s interior converge on core Ts of 4000°C ± 500 °C Thermal energy moves from hot to cold--> thus, modes of energy transport within Earth: • Conduction • Convection • Radiation
How do we know that convection is important? Thought experiment: Distance heat transported by conduction = sqrt (thermal diffusivity * age of Earth) • Thermal diffusivity = 10-6 m2/s • 3.2 x 107 sec/yr
How do we know that convection is important? 10-6 m2/s * 4.5 x 109 yr * 3.2 x 107 sec/yr = 380 km Radius of Earth = 6371 km Conclusion: barely any heat transported by conduction. Requires a convective mechanism.
models from: http://www.geo.lsa.umich.edu/~crlb/COURSES/270 convection in the mantle observed heat flow warmer: near ridges colder: over cratons from: http://www-personal.umich.edu/~vdpluijm/gs205.html
examples from western Pacific blue is high velocity (fast) …interpreted as slab note continuity of blue slab to depths on order of 670 km from: http://www.pmel.noaa.gov/vents/coax/coax.html
Volcano geography 1. Divergent margins 2. Convergent margins 3. Intraplate 4. Hotspots
Plate tectonics and magma composition 1. Divergent margins: Plate separation and decompression melting -> low volatile abundance, low SiO2 (~50%), low viscosity basaltic magmas (e.g. Krafla, Iceland) 2. Convergent margins : Mixtures of basalt from the mantle, remelted continental crust and material from the subducted slab. High volatile abundance, intermediate SiO2 (60-70%), high viscosity andesites and dacites (e.g. Montserrat, West Indies) 3. Intraplate `Hot-spot` settings: A. Oceanic: Mantle plumes melt thin oceanic crust producing low viscosity basaltic magmas (e.g. Kilauea, Hawaii) B. Continental: Mantle plumes melt thicker, silicic continental crust producing highly silicic (>70% SiO2) rhyolites (e.g. Yellowstone, USA)
What are the plate tectonic settings in which magmatism occurs?