chapter five: microbial metabolism
chapter five: microbial metabolism. oxidation-reduction. redox reaction: coupled reactions. e - removed as part of H atom. redox reactions. aerobic respiration. oxygenic photosynthesis. carbon source organic vs. CO 2. heterotroph. autotroph. energy source light/chemical.

chapter five: microbial metabolism
E N D
Presentation Transcript
oxidation-reduction redox reaction: coupled reactions e- removed as part of H atom
redox reactions aerobic respiration oxygenic photosynthesis
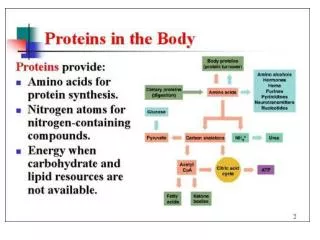
carbon source organic vs. CO2 heterotroph autotroph energy source light/chemical energy source light/chemical chemoautotroph photoheterotroph chemoheterotroph photoautotroph electron source organic/inorganic electron source organic/inorganic electron source organic/inorganic electron source inorganic chemoorgano- autotroph chemolitho- autotroph photoorgano- heterotroph photolitho- heterotroph photolitho- autotroph chemoorgano- heterotroph chemolitho- heterotroph O2 vs. inorganic electron acceptor organic/inorganic fermentation respiration O2 vs. other oxygenic photosynthesis anoxygenic photosynthesis aerobic respiration anaerobic respiration nutritional classification: metabolic strategy sulfur oxidizers iron oxidizers lactic acid alcoholic mixed acid butanediol nitrate carbonate sulfate
complementary metabolism autotrophy heterotrophy
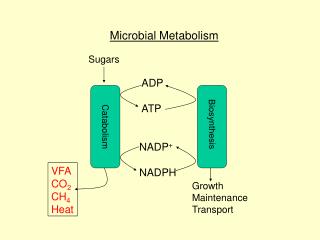
C6H12O6 NAD+ ETC NADH CO2 ADP + P ATP 2 H+ heterotrophy: respiration NO2-, N2 + H2O H2S + H2O CH4 + H2O H2O NO3- SO4- CO32- O2 cell material
C6H12O6 C6H12O6 NAD+ NAD+ ETC NADH NADH CO2 substrate P few ATP pyruvate ADP + P lots of ATP 2 H+ heterotrophy: respiration & fermentation reducede- acceptor inorganic e- acceptor lactic acid ethanol & CO2 mixed acids butanediol organic pyruvate ferm cell material
heterotrophy: respiration & fermentation respiration • inorganic e- acceptor • does NOT mean O2 • organic mole. CO2 fermentation • organic e- acceptor • organic organic mole. • incomplete H stripping,lower ATP yield
Chapter Five Learning Objectives • Discuss redox reactions in biological systems. • Identify the redox partners in aerobic and anaerobic respiration and oxygenic and anoxygenic photosynthesis. • Correctly identify the carbon, energy and electron source for an organism when given its nutritional classification (e.g., chemoorganoheterotroph). • How is ATP generated in both substrate level and oxidative phosphorylation? • Why is it so important that the electron transport chain is housed in a lipid bilayer membrane? Why is a terminal electron acceptor so important? • What happens in a microorganism if the terminal electron acceptor of the ETC is not available? What molecules build up? What is done with these molecules? • How do amphibolism, catabolism and anabolism relate to growth and repair in cells? • Discuss the major differences between respiration and fermentation. What are the four basic kinds of fermentation?
chemo-: conversion of chemical E ATP sulfur oxidation iron oxidation -synthesis: carbon fixation (CO2 organic molecule) autotrophy: chemosynthesis NAD+ NADH NAD+ NADH ETC ETC carbon fixation carbon fixation ADP + P ADP + P ATP ATP 2 H+ H2S SO42- 2 H+ 2Fe2+ 2Fe3+
chemosynthesis: iron oxidation Thiobacillus ferrooxidans chemolithoautotrophy • energy = Fe2+ Fe3+ • electron = same • carbon = CO2 CH2O
chemosynthesis: sulfur oxidation Sulfolobus acidocaldarius chemolithoautotrophy • energy = S2- (sulfide) / S2O32- (thiosulfate) SO32- (sulfite) • electron = same • carbon = CO2 CH2O
autotrophy: photosynthesis photo: light E chemical E light-dependent (light) reactions ATP & NAD(P)H “reducing power” synthesis: light-independent (dark) reactions carbon fixation: piling e- onto CO2 NAD(P) NAD(P)H chlorophyll ETC carbon fixation (light independent reactions) oxidized chlorophyll ADP + P ATP H2S/H2O
photosynthetic electron flow & chemiosmosis cyclic photosynthesis in the purple sulfur bacteria non-cyclic photosynthesis in the cyanobacteria
photosynthesis compared [O2] [H2S]
chapter 5 learning objectives • How is ATP generated in chemosynthesis, photosynthesis and respiration? How is the process different for each and how is it the same? • Discuss the redox partners of sulfur and iron oxidizing bacteria. • How do non-cyclic and cyclic photosynthesis differ? How does each produce ATP and NADPH/NADH? What is each used for? • How is carbon fixed during chemosynthesis and photosynthesis? How is the process similar and how is it different?