BioTech
David: BioTech Industry Dave: Amgen Christine: Genentech Karen: Protein Design Labs. BioTech. 2000 TV AD. 2004 TV AD. Definition. Sector: HealthCare Industry: Biotechnology & Drugs

BioTech
E N D
Presentation Transcript
David: BioTech Industry Dave: Amgen Christine: Genentech Karen: Protein Design Labs BioTech
Definition Sector: HealthCare Industry: Biotechnology & Drugs Definition: engaged in the discovery, development, and commercialization of biotechnology based drugs and products (therapeutic proteins, recombinant DNA vaccines, gene therapies, and devices).
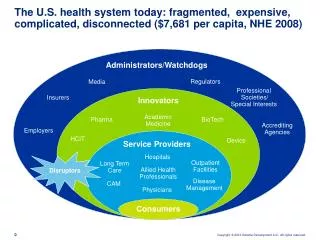
Industry Positioning Amgen, Genentech Size Protein Design Labs Business Function
The Market Cancer • about 1 in 3 lifetime risk; 38% of women and 43% of men • 23 million suffering worldwide. • Estimated $280 billion spent on treatment drugs for cancer annually. Diabetes • 16 million Americans with 10.3 million diagnosed and 8.1 million women (NWHIC); 65 per 1000 - NHIS95; 8 million - perhaps 16 million if include not-yet-diagnosed. • 165 million cases worldwide (2003) • $150 billion in direct and indirect costs in America Heart Disease • 25 million adults in the US • Heart disease and stroke cost US almost $260 billion annually.
Recent BioTech Timeline 4000–2000 B.C. Biotechnology first used to leaven bread and ferment beer, using yeast (Egypt). 1990 • Chy-Max™, an artificially produced form of the chymosin enzyme for cheese-making, is introduced. It is the first product of recombinant DNA technology in the U.S. food supply. • The first experimental gene therapy treatment is performed successfully on a 4-year-old girl suffering from an immune disorder. 1993 FDA approves bovine somatotropin (BST) for increased milk production in dairy cows. 1994 First FDA approval for a whole food produced through biotechnology: FLAVRSAVR™tomato. 1997First animal cloned from an adult cell: a sheep named Dolly in Scotland. 1998 A rough draft of the human genome map is produced, showing the locations of more than 30,000 genes. 1999 Biotechnology-based biopesticide approved for sale in the United States. 2001 Chinese National Hybrid researchers report developing a "super rice" that could produce double the yield of normal rice. 2002 The draft version of the complete map of the human genome is published. 2003 • GloFish, the first biotech pet, hits the North American market. Specially bred to detect water pollutants, the fish glows red under black light thanks to the addition of a natural fluorescence gene. • The U.K. approves its first commercial biotech crop in eight years. The crop is a biotech herbicide-resistant corn used for cattle feed. • Japanese researchers develop a biotech coffee bean that is naturally decaffeinated. 2004 The FDA approves the first anti-angiogenic drug for cancer, Avastin (bevacizumab).
Industry Facts • Biotechnology is responsible for hundreds of medical diagnostic tests that keep the blood supply safe from the AIDS virus and detect other conditions early enough to be successfully treated. Home pregnancy tests are also biotechnology diagnostic products. • Consumers already are enjoying biotechnology foods such as papaya, soybeans and corn. • Environmental biotechnology products make it possible to clean up hazardous waste more efficiently. • Industrial biotechnology applications have led to cleaner processes that produce less waste and use less energy and water in such industrial sectors as chemicals, pulp and paper, textiles, food, energy, and metals and minerals. • DNA fingerprinting, a biotech process, has dramatically improved criminal investigation and forensic medicine, as well as afforded significant advances in anthropology and wildlife management. • There are 1,473 biotechnology companies in the United States, of which 314 are publicly held. • Market capitalization: $311 billion as of mid-March 2004. • Revenues: increasing from $8 billion in 1992 to $39.2 billion in 2003. • Employed: 198,300 peopleas of Dec. 31, 2003. • R & D: spent $17.9 billion on research and development in 2003. • The top eight biotech companies spent an average of $104,000 per employee on R&D in 2003. • Regulated: by the U.S. Food and Drug Administration (FDA), the Environmental Protection Agency (EPA) and the Department of Agriculture (USDA). • There are more than 370 biotech drug products and vaccines currently in clinical trials targeting more than 200 diseases, including various cancers, Alzheimer's disease, heart disease, diabetes, multiple sclerosis, AIDS and arthritis.
6 Key Investment Guidelines • Product Pipeline Look for companies with at least two drugs in clinical trials • Collaborations Collaborative agreements with various pharmaceutical companies for research or marketing and link up with a corporate or academia partner => lower risk • Management Proven track record of taking a drug through the regulatory hurdles and/or to the marketplace • Cash Company’s burning of cash in ongoing research and development or "burn rate" is a critical measure of a company’s longevity • Market Aimed at markets for a new drug that is both large and under-serviced. • Technology Look to see if the research and development can do what it needs to do and supposedly solve a medical problem or problems.
FDA Food andDrug Administration • Sets health and safety standards • Drugs, food , medical devices, cosmetics products, and biologics • Also monitor for proper production standards • Ensure labeling is truthful and informative.
Pre Clinical Tests Tests on • Isolated tissues • Cell Cultures • Animals To see the potential effects on humans • The beginning of the drug approval process • Company decides whether to pursue human testing
The IND Filing • Exemption from marketing application in order to transport the drug • Before testing on humans can begin, the company must file an Investigational New Drug (IND) application • FDA has 30 days to review the IND application • At this time patents are usually applied for, patents last generally for 20 years • The goal of the IND application is to provide pre-clinical data of sufficient quality to justify the testing of the drug in humans • About 85% of all IND applications move on to begin clinical trials
Patent • Biotech inventions are subject to the same rules as all other inventions • Generally last 20 years • What can be patented • Product • Method • Use • Examples • DNA and RNA sequences • proteins, enzymes, antibiotics • antibodies, antigens • micro-organisms, cell lines, hybrids
Phase I • Ranging from 1 to 3 years • Begins to analysis the drugs safety profile • How the drug is absorbed, metabolized and excreted • Improves Chances of making it to market by 30% • Sample in this phase is small (less than 100 patients) • Healthy volunteers => safety and dosage
Phase II • Ranging about 2 years • Consists of small, well-controlled experiments to evaluate the drug's safety and assess side effects • Making it through phase two increase your chances of being approves by 60% • Sample size of 100 – 300 patients who suffer from the disease • This phase establishes the optimal dosage of the drug
PhaseIII • Ranging between 3 and 4 years • Verifies the drug is effective in its intended use • Assessment of long term effects • Continues to build the safety profile of the drug and record possible side effects and adverse reactions resulting from long-term use • Sample size of at least 1000 patients • At this point the drug has a 70% chance of FDA approval. Processing of the NDA New Drug Application • This can take be done in as little as 6 month or as long as 2 years • Drugs are subject to ongoing review, making sure no adverse side effects appear from the drug.
Phase IV • Observational studies in an ongoing evaluation of the drug's safety during routine use • Monitor any usage of the drug for conditions other than the approved medical indication
Key Points • The clinical trial process is costly as well as time-consuming • $350 - $500 million to bring a drug through all phases of the clinical trial process • Consider companies that already have: • one or more successful products on the market • a large pipeline of candidate drugs • plenty of cash to fund the development of their new drug candidates
Porter’s Competitive Force LOW MID LOW LOW-MID LOW-MID
Critical Challenges Political & Legal: • Product liability • Pressure from governments worldwide to contain drug prices. Social: • Ethical concerns over controversial aspects of its research.
The Future Toward profitability by the end of the decade. • rebounded in dramatic fashion in 2003 and 2004 from a stock market decline • moving new, first-in-class medicines through clinical trials and onto the market. • we predict the industry should achieve the first net income in its 30-year history in 2008. From potential to performance. • real products—life-saving medicines awaiting approval by the U.S. Food and Drug Administration (FDA), or in late-stage clinical trials. • The biotech industry has moved from technology-driven to product-driven, from potential to performance. With a robust pipeline of products, the industry is on track year-after-year to set new standards of medical care and revenue growth. • Biotech has achieved a critical mass of products. Companies are judged by higher sales, first-in-class approvals, and ground-breaking successes in Phase II and Phase III trials. Emerging technologies are valued for their contributions in creating products in the near-term.
The Future The future is bright. • biotech industry has reached a critical mass of products on the market and in late-stage clinical trials, driving its performance to a higher level of stability and predictability. Biotech is an industry of fundamentally sound companies creating essential products for improving the health and quality of life of everyone on the planet. With its recent resurgence, the industry’s best days are still to come.
It starts with a f lash of insight— a potentially life-changing discovery.
Business Profile • Pioneers of the development of innovative products based on advances in recombinant DNA and molecular biology • Human Therapeutics • Oncology (supportive cancer care) • Inflammation (rheumatoid arthritis) • Hematology & nephrology (anemia, kidneys) • Neurology (brain, nervous system) • Metabolic Disorders (autoimmune diseases, viruses)
Mission Statement Mission Statement: “At Amgen, our mission is to serve patients. As a science-based, patient-focused organization, we will follow the science wherever it may lead, to discover and develop innovative therapies to treat grievous illness.”
Organizational Values Be Science BasedCompete Intensely and WinWork in Teams Create Value for Patients, Staff and Stockholders Trust and Respect Each OtherEnsure Quality Collaborate, Communicate and Be AccountableBe Ethical
Company Strategy • Principal products marketed to clinics hospitals and pharmacies, big 4 products are also marketed outside the US. Outside the US products marketed mostly to wholesalers or hospitals depending on distribution practice in that country. • Research facilities in US, clinical development staff in Europe Canada Australia and Japan • Also utilize R&D collaborations and licensed certain product and technology rights to enhance R&D efforts • Manufacturing facilities in US Puerto Rico and Netherlands, also have 3rd party contract manufacturers. • Overall,a big time industry leader that focuses on research and utilizes strategic alliances and acquisitions
Some Quick Facts • Headquarters: Thousand Oaks, California • Stock Information: • Nasdaq: AMGN • Number of Staff Members: Nearly 13,000 (as of year-end 2003) • Chairman and CEO: Kevin W. Sharer
History of Amgen • 1980 - Company founded, financed by venture capitalists Bill Bowes, Frank Johnson, Sam Wohlsteader, and Raymond Baddour • 1983 - IPO of 2,350,000 shares @ $18 • erythroprotein first cloned- later becomes drug epogen • 1987 - Co’s name changed to Amgen, becomes a Delaware corporation • 1989 - Immunex clones TNF receptor, later patented and named -ENBREL®Amgen receives first U.S. patent for recombinant G-CSF (NEUPOGEN®)FDA approves EPOGEN® for the treatment of anemia in patients with end-stage renal diseaseEPOGEN® named "Product of the Year" by Fortune magazineAmgen added to the NASDAQ 100 Index
History • 1991 - FDA approves NEUPOGEN® to decrease the incidence of infection associated with chemotherapy-induced neutropenia in patients with non-myeloid cancers receiving myelosuppressive therapy • 1996 - FDA approves EPOGEN® for reduction of allogeneic blood transfusion in surgery patientsAmgen reaches sales of more than $2 billion • 1998 - FDA approves ENBREL® to treat patients with rheumatoid arthritis whose disease has failed to respond to prior disease-modifying medicines • 2000 - Kevin W. Sharer named Chief Executive OfficerAmgen acquires Kinetix Pharmaceuticals