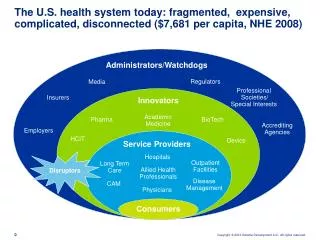
Trinity Biotech
Trinity Biotech . Fingerstick whole blood sample for HIV OTC test Presented by; Fiona Campbell. Trinity experience with rapid HIV testing. Commercializing HIV rapid tests internationally for more than 12 years

Trinity Biotech
E N D
Presentation Transcript
Trinity Biotech Fingerstick whole blood sample for HIV OTC test Presented by; Fiona Campbell
Trinity experience with rapid HIV testing • Commercializing HIV rapid tests internationally for more than 12 years • In the last year alone over 7 million persons have been tested for HIV by Trinity Biotech Plc. rapid HIV products • Different test formats • Varied users and applications
PMA approval 23rd December 2003 Approved for use with serum, plasma and whole blood (fingerstick and venipuncture) CLIA waived for venipuncture whole blood and fingerstick whole blood UniGoldTM Recombigen HIV
User experience with Fingerstick Whole Blood • Glucose monitoring, Cholesterol monitoring, Hemoglobin, Lactic acid, Triglycerides • FDA approved HIV home test (Home Access) • Low virus viability ex-vivo • Safe disposal of bloods can be easily achieved • Sealed disposal containers • Sterile disinfectant wipes • Retractable safety lancet
Procedure Read instructions Clean finger – sterile wipe Prick finger – retractable safety lancet Take blood sample - measured capillary tube Add blood to device Add buffer x 4 drops Wait 10 minutes Interpret result Dispose of materials Ease of Use
2 built in controls Sample addition Test functionality User Safety Mechanisms
Test Principals and Performance data • Third-generation, antigen sandwich format • Allows for earlier detection of antibodies during sero-conversion phase • Sensitivity (whole blood samples) • 100% 1032/1032 95% C.I. = 99.5-100.0% • Specificity (whole blood samples) • Prevalence >1% 99.8% (95% C.I. 99.2-100%) • Prevalence <1% 99.7% (95% C.I. 99.0-100%) • Sero conversion and low titre panels • The UniGold test showed comparable sensitivity to the most sensitive FDA licensed EIA tests.
CLIA waiver data / Untrained user • Testing of blinded panel • 100 untrained subjects at 3 sites • Interpretation of picture cards • 60 untrained subjects interpreted 8 pictures • Flex / tolerance studies both in house and external studies
Summary • OTC use with whole blood is established • The UniGoldTM Recombigen HIV is a suitable format for OTC use • Trinity Biotech is committed to supporting international use of HIV rapid tests.