Molecules and Compounds (Nomenclature) Chapter 5
430 likes | 595 Views
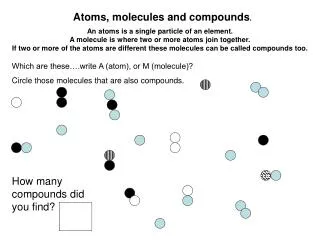
Molecules and Compounds (Nomenclature) Chapter 5. Tro, 2 nd ed. Molecules and Compounds. Pair up and look up this law and then write it in your own words to share with the rest of the class. Give examples other than what is in the textbook. Law of Constant Composition.

Molecules and Compounds (Nomenclature) Chapter 5
E N D
Presentation Transcript
Molecules and Compounds(Nomenclature)Chapter 5 Tro, 2nd ed.
Molecules and Compounds Pair up and look up this law and then write it in your own words to share with the rest of the class. Give examples other than what is in the textbook. Law of Constant Composition
HOW TO LEARN NOMENCLATURE Memorize, memorize, memorize! Memorize the rules for naming Memorize names of ions Memorize some acid names and some organic names
Types of Compounds and Naming Rules Compound Ionic (formula unit) Covalent (molecule) Salts Hydrates Other Binary (Ternary) Organic Acids Other Binary Polyatomic Ions You will learn all the rules for ionic, hydrates, covalent binary, and only specific common names, organic compounds and acids.
Common and Systematic Names Chemical nomenclature is the system of names that chemists use to identify compounds. Two classes of names exist: common names and systematic names. Common names are arbitrary names. They are not based on the composition of the compound. They are based on an outstanding chemical or physical property. Common names that you need to memorize: acetylene, ammonia, water, baking soda, cane sugar, epsom salts, grain alcohol, laughing gas, lye, muriatic acid, table salt, vinegar, washing soda, and wood alcohol. (common name, formula and systematic name)
Chemists prefer systematic names. Systematic names precisely identify the chemical composition of the compound. The present system of inorganic chemical nomenclature was devised by the International Union of Pure and Applied Chemistry (IUPAC).
Sodium Na Potassium K Zinc Zn Argon Ar Mercury Hg Lead Pb Calcium Ca The formula for most elements is the symbol of the element.
These 7 elements are found in nature as diatomic molecules. Hydrogen H2 Nitrogen N2 Oxygen O2 Fluorine F2 Chlorine Cl2 Bromine Br2 Iodine I2
If one or more electrons are removed from a neutral atom a positive ion is formed. A positive ion is called a cation. A charged particle known as an ion can be produced by adding or removing one or more electrons from a neutral atom. remove e- → neutral atom cation
MONATOMIC IONS: Metals always lose electrons to form positive ions, called cations If element forms only one cation, we name it by its element name: Na+ = sodium ion Al3+ = aluminum ion If a transition metal can form more than one cation, add ion’s charge in Roman numerals to the name. Fe2+ is iron(II) ion and Fe3+ is iron(III) ion. (Old names, ferrous and ferric.)
If one or more electrons are added to a neutral atom a negative ion is formed. A negative ion is called an anion. A charged particle known as an ion can be produced by adding or removing one or more electrons from a neutral atom. add e- → neutral atom anion
MONATOMIC IONS: Nonmetal atoms gain electrons to form negative ions, called anions Take the root of the element name and add “-ide” Cl- = chloride ion Se2- = selenide ion N3- = nitride ion Most often ions are formed when metals combine with nonmetals. (Exception is NH4+ with anion.) The charge on an ion can be predicted from its position in the periodic table.
elements of Group VIIA have a-1 charge elements of Group VIA have a -2 charge elements of Group VA have a-3 charge elements of Group IA have a +1 charge elements of Group IIA have a+2 charge
NAMING IONIC COMPOUNDS: Always put cation before anion in formula. Name of compound is just cation name followed by anion name. MgF2 is Mg2+ and F- = magnesium fluoride FeBr3 is Fe3+ and Br- = iron(III) bromide What is the formula for silver carbonate? Silver ion is Ag+ and carbonate CO32- - have to balance charges, so need 2 Ag+ for each CO32- = Ag2CO3 Generic: Ma+bNb-a criss-cross rule However, if a = b, then both = 1 by definition for lowest ratio Figure out the charge on each x: ZnX, NH4X, (NH4)2X, Al2X3, X2(SO3)3
Write the formula of barium phosphide. The lowest common multiple of +2 and –3 is 6 Step 1. Write down the formulas of the ions. Ba2+P3- Step 2. Combine the smallest numbers of Ba2+ and P3- so that the sum of the charges equals zero. The cation is written first. The anion is written second. 3(Ba2+) +2(P3-) = 0 3(2+) + 2(3-) = 0 The correct formula is Ba3P2
Naming Compounds Containing Polyatomic Ions A polyatomic ion is an ion that contains two or more elements. Compounds containing polyatomic ions are composed of three or more elements. They usually consist of one or more cations combined with a negative polyatomic ion. When naming a compound containing a polyatomic ion, name the cation first and then name the anion.
The ions are what isactually present. This is the way theformula is written.
phosphite phosphate nitrite nitrate Elements that Form More than One Polyatomic Ion with Oxygen Anions ending in -atealways contain more oxygen than ions ending in -ite.
One group of ionic compounds that contains all nonmetals is the ammonium salts. Try to write formulas for ammonium sulfate and ammonium phosphate.
ANIONS WITH HYDROGEN Some anions have picked up one or two hydrogen ions. Old naming rules put “bi” in front of the anion name. IUPAC uses hydrogen or dihydrogen: CO32- with one H+ added is HCO3- Carbonate ion becomes hydrogen carbonate ion. Name these: PO43-, HPO42-, H2PO4-
hydroxide cyanide hydrogen sulfide peroxide Four polyatomic ions that do not use the –ate/ite system.
mercury(I) ammonium hydronium There are three common positively charged polyatomic ions.
Divide the formula into cation(s) and anion. Name each. For example KHSO4 is K+ and HSO4-. MgNH4PO4 is Mg2+, NH4+ and PO43-.
Binary Compounds Binary compounds contain only two different elements. There are four main types, listed in following slides. • Binary Ionic Compounds Containing a Metal Forming Only One Type of Cation (salts) Binary ionic compounds consist of a metal combined with a non-metal. The chemical name is composed of the name of the metal followed by the name of the nonmetal which has been modified to an identifying stem plus the suffix –ide. Using this system the number of atoms of each element present is notexpressed in the name. Practice: CaC2, MgBr2, Al2O3, NaH (calcium carbide, magnesium bromide, aluminum oxide, sodium hydride)
Binary Compounds B. Binary Ionic Compounds Containing a MetalThat Can Form Two or More Types of Cations (still salts) The chemical name is composed of the name of the metal with its charge in Roman numerals, followed by the name of the nonmetal which has been modified to an identifying stem plus the suffix –ide. Using this system the number of atoms of each element present is not expressed in the name. Practice: FeS, CuCl2, SnF2, Mn3(PO4)5 (iron (II) sulfide, tin(II) fluoride, manganese (V) phosphate)
Ion Names: Classical System 2+ 2 Lower Charge Higher Charge This is for information and for homework only; classical names will not be on quiz or test unless part of a common name that’s required.
Binary Compounds C. Binary Compounds Containing Two Nonmetals (Binary Covalent) Compounds between nonmetals are molecular, not ionic. In a compound formed between two nonmetals, the element that more electropositive (least electronegative) is named first. Rules for naming: 1. Give Greek prefix (to indicate number of atoms of first element) to name of first element. Don’t use mono prefix for first element. 2. Give Greek prefix (to indicate number of atoms of second element) to root of element name, then add -ide Example: N2O3 is dinitrogen trioxide. Exception: hydrogen never has prefix.
Greek Prefixes mono = 1 di = 2 tri = 3 tetra = 4 penta = 5hexa = 6 hepta = 7 octa = 8 nona = 9 deca = 10
Binary Compounds C. Binary Compounds Containing Two Nonmetals (Binary Covalent)Examples N2O3, PCl5, Cl2O7, CCl4, CO, CO2, PI3, H2S Dinitrogen trioxide Phosphorous pentachloride (not mono-) Dichlorine heptoxide Carbon tetrachloride Carbon monoxide Carbon dioxide Phosphorous triiodide Hydrogen sulfide (hydrogen never gets a prefix) Now try: CS2, H2O, N2O4, SF2, SiCl2, P2O5, phosphorous trichloride, dichlorine oxide
Binary Compounds D. Acids Derived from Binary Compounds Certain binary hydrogen compounds, when dissolved in water, form solutions that have acid properties. The aqueous solutions of these compounds are given acid names. The acids names are in addition to their –idenames. Hydrogen is typically the first element of a binary acid formula.
binary hydrogen compound like HCl is called hydrogen chloride (not an acid yet). Acid sol. water Acid Formation Now it’s hydrochloric acid.
Binary Compounds D. Acids Derived from Binary Compounds To name binary acids write the symbol of hydrogen first. After hydrogen write the symbol of the second element. Place the prefix hydro- in front of the stem of the nonmetal name. Place the suffix -ic after the stem of the nonmetal name. Practice: HCl, H2S, HI (hydrochloric acid, hydrosulfuric acid, hydroiodic acid)
D. A. C. B.
The other element is usually a nonmetal, but it can be a metal. The first element listed in the formula is hydrogen. The remainingelements include oxygen and form a polyatomic ion. The word acid in the name indicates the presence of hydrogen. Oxy-acids contain hydrogen, oxygen and one other element. Hydrogen in an oxy-acid is not expressed in the acid name. More Acids
indicates hydrogen contains hydrogen contains sulfur contains oxygen sulfuric acid This is one of the six acids you have to know.
(If you add HCl to the ones marked by arrows, you have your total list of acids.)
Your Acids An acid is a substance that forms H+ when dissolved in water Some are strong acids, that means all the H+ dissolves and dissociates from the anion Some acids are weak, meaning very little H+ dissociates, although the cmpd dissolves Forget sections in text: just learn these! Hydrochloric acid, sulfuric acid, and nitric acid are strong acids: HCl(aq), H2SO4(aq), HNO3(aq) Acetic acid, carbonic acid, phosphoric acid are weak acids: HC2H3O2(aq), H2CO3(aq), H3PO4(aq)
Your Organic Compounds No naming rules – just learn formulas and names. CH4 methane, C3H8 propane, C8H18 octane, CH3OH methanol, CH3CH2OH ethanol, C6H6 benzene, CH3COOH acetic acid (organic method), CH3NH2 aminomethane, C6H12O6 glucose, C12H22O11 sucrose, and C2H2, ethyne
HYDRATES: (not in book) Also a good review of ionic names! If an ionic compound is a hydrate, it will have *H2O in the formula, like MgCO3*6 H2O Name the ionic compound part using ionic naming rules, then give the Greek prefix for the number of water molecules and add the word hydrate: MgCO3*6H2O is magnesium carbonate hexahydrate
HYDRATES: (not in book) Also a good review of ionic names! Examples: you name the following (notice their common names) gypsum CaSO4*2 H2O epsom salts MgSO4*7 H2O washing soda Na2CO3*10 H2O bluestone CuSO4*5 H2O Calcium sulfate dihydrate Magnesium sulfate heptahydrate Sodium carbonate decahydrate Copper(II) sulfate pentahydrate
Formula Mass As each element’s atom has mass in amu on the Periodic Table, so do molecules and formula units. Formula mass is the sum of the atomic masses of the atoms in the chemical formula. Practice: water, sodium chloride, acetic acid