Compounds and Molecules
Compounds and Molecules. 4.1. vid. What are compounds?. Two or more elements chemically combined to form new substances. What are compounds?. Chemical Bonds – the attractive forces that hold different atoms or ions together Bonds distinguish compounds from mixtures. Chemical Formula.

Compounds and Molecules
E N D
Presentation Transcript
Compounds and Molecules 4.1 vid
What are compounds? • Two or more elements chemically combined to form new substances.
What are compounds? • Chemical Bonds – the attractive forces that hold different atoms or ions together • Bonds distinguish compounds from mixtures.
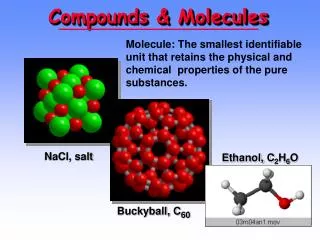
Chemical Formula • C12H22O11 can only be sugar • H20 can only be water
Parts to the Formulas 2H2O Subscript Coefficient
Parts to the Formulas • Coefficient – gives the amount of molecules or moles • Subscript – tells the number of atoms or ions
Chemical Structure • Bond length -the distance between two bonded atoms • Bond angle -the angle formed between two bonded atoms
Models of compounds • Ball and stick model -atoms are represented balls, bonds are represented by sticks. H H BondAngle O Bond length
Models of compounds • Structural formulas -chemical symbols represent the atoms. H H O
Models of compounds • Space-filling -show the space occupied by each atom represented in the compound. Hydrogen Oxygen
Structure of Compounds • Network Structures = strong solids • Molecules= weak solids, liquids, or gases
Ionic and Covalent Bonding 4.2 fun
Why Bond? • Atoms bond so they can become stable! • They want to achieve a full outer energy level • They want 8 electrons.
1. Ionic bond • A bond formed by the attraction between oppositely charge ions. • The joining of a positive metal ion and a negative nonmetal ion Na+ + Cl- NaCl
Ionic bond • Formed by transferring electrons • Now each ion will have 8 valence electrons NaCl Na+ + Cl-
Ionic bond Properties • They form network structures • high melting points • Good conductors of electricity when dissolved or melted because of their ions
Metallic bond • Formed by the attraction between positively charged metal ions and the electrons around them.
Metallic bond • Held together by free flowing electrons • Hold metallic elements together
Covalent Bonds • Formed when atoms share one or more valence electrons • Formed between non-metals • May form single, double, or triple bonds
Covalent Properties • They are poor conductors of electricity (no ions) • Low melting points. • form molecules
Polyatomic ion • An ion made of two or more atoms that are covalently bonded and that act like a single ion. • A molecule with a charge • Examples: carbonate (CO3)-2 Nitrate (NO3)-1
REMEMBER!!!!!!!! • If it starts with metal it is an ionic compound • If it starts with a nonmetal it is a covalent compound • If the formula contains a parentheses (polyatomic ion) it contains both ionic and covalent bonds • If it is just a metal it is metallic
Writing Ionic Formulas Calcium Fluoride Step 1 - Look up both positive and negative ions Ca2+ F-1
Writing Ionic Compounds Step 2 Criss-cross numbers down, making subscripts Ca2+ F-1 Ca1 F2
Writing Ionic Compounds Step 3 Combine your new compound. Ca1 + F2 Ca1F2
Polyatomic ion • Al+3 (CO3)-2 • Al2(CO3)3 • Al2(CO3)3 Aluminum Carbonate
Naming Ionic Compounds • Step one – Name the first ion in the formula Ex. CaCl2 – calcium Na2(SO4) - sodium
Naming Ionic Compounds • Step two – Name the second ion Ex. CaCl2 –Calcium Chloride Na2(SO4)-sodium sulfate
PROBLEM Ex. Fe2O3 and FeO are different compounds from different ions • Fe3+ O2- = Fe2O3 • Fe2+ O2- = FeO
Solution-Roman Numerals • Same naming scheme • Add a Roman Numeral to identify the charge of the metal ion.
Solution-Roman Numerals Fe2O3 = iron(III)Oxide FeO = iron(II)Oxide • Remember!!! • R.N. is the charge, not the subscript of the metal.
Mono – 1 Di - 2 Tri - 3 Tetra - 4 Penta - 5 Hexa – 6 Hepta - 7 Octa - 8 Nona - 9 Deca - 10 Naming Covalent Compounds
Naming Covalent Compounds • Step 1 Identify the number of atoms in the first element Ex. H2O two H atoms
Naming Covalent Compounds • Step 2 Identify the number of atoms in the 2nd element. Ex. H2Oone O atom
Naming Covalent Compounds • Step 3 Name the compound with the appropriate prefixes. Ex. H2O dihydrogen monoxygen
Naming Covalent Compounds • Step 4 add an -ide ending to the second element Ex. Dihydrogen monoxide
Empirical Formula • The simplest chemical formula of a compound. • Different covalent compounds can have same empirical formula