Partial Pressure
Partial Pressure. Primary determinant of diffusion and direction Describes the pressure of a particular gas within a mixture Equals the total pressure times the fractional concentration of the particular gas, according to Whose Law?. Partial Pressure. Concentration of gases in ambient air

Partial Pressure
E N D
Presentation Transcript
Partial Pressure • Primary determinant of diffusion and direction • Describes the pressure of a particular gas within a mixture • Equals the total pressure times the fractional concentration of the particular gas, according to Whose Law?
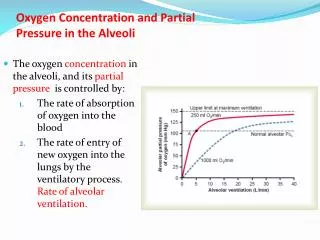
Partial Pressure • Concentration of gases in ambient air • Oxygen 20.93% x PB @ sea level = 159 mm Hg • Nitrogen 79.04% x PB = 600.7 mm Hg • Carbon dioxide 0.03% x PB = 0.2 mm Hg • Tracheal air becomes diluted w/ water vapor • 760 – 47 = 713 mm Hg • 713 x .2093 = 149 mm Hg PO2 • Alveolar air is mixed with CO2 leaving blood • Oxygen concentration = 14.7% (x 713 = 104 mm Hg PO2)
Partial Pressure Alveolar Air Percentages and Partial Pressures differ from Ambient Air and Tracheal Air Humidification reduces pressure of gasses Oxygen is constantly leaving alveoli in capillaries Some residual air is always left in alveolus in each breath to mix with new air.
Gas Transport External respiration Oxygen moves from alveoli into blood down concentration gradient (104 mm Hg to 40 mm Hg. Carbon dioxide moves from blood into alveoli down concentration gradient (46 – 40 mm Hg). Internal respiration Carbon dioxide produced in cells moves from higher pressure to lower pressure into blood.
Oxygen Transport in the Blood • Dissolved in plasma • amount is proportional to partial pressure • O2 is poorly soluble in plasma • average .3 ml O2 / 100 ml blood • Combined with hemoglobin • each hemoglobin molecule combines with 4 O2 • average 20 ml O2 / 100 ml blood
Oxygen Capacity of Hemoglobin • Men average grams of hemoglobin = 15/100 ml • Women average grams of hemoglobin = 14/100 ml • Amount of O2 per gram of hemoglobin = 1.34 ml • Maximal amount of O2 that combine 100 ml blood = • 20.1 ml O2 for men • 18.8 ml O2 for women if 100% saturated.
Oxygen Capacity of Hemoglobin • Percent saturation (%SO2) is proportion of hemoglobin bound with oxygen. • Even with sufficient atmospheric oxygen, adequate pulmonary ventilation, & optimum diffusing capacity, Hb not become 100% saturated. • SO2 arterial blood at rest = 98% • Hemoglobin carries 19.7 ml O2 (.98 x 20.1) and .3 ml dissolved in 100 ml blood at 100 mm Hg as in the lungs.
Oxygen Capacity of Hemoglobin • Tissue PO2 in cell fluids at rest averages 40 mm Hg. • Dissolved oxygen in arterial blood readily diffuses into cells. • This causes Hb to release its oxygen. • At 40 mm Hg, Hb holds about 75% of total capacity for oxygen (15 mL O2). • Arteriovenous-oxygen difference at rest = 5 mL O2
Hb Saturation Differences in Arteries and Veins Arteriovenous O2 difference • Reveals the amount of oxygen extracted by the tissues
Oxygen Capacity of Hemoglobin • During exercise, tissue PO2 decreases to about 20 mm Hg. • Hemoglobin retains about 25% or 5 mL O2 (.25 x 20.1). • A-v O2 difference = 20.1 – 5 = 15 mL/100. • During exhaustive exercise, tissue PO2 about 3 mm Hg, Hb releases all.
Blood Conditions Influencing O2 Saturation of Hemoglobin • Temperature • pH • PCO2 • PO2 • 2,3-diphosphoglycerate (also called 2,3-DPG or BPG)
Resting Conditions • Temperature = 37 degrees Celsius • pH = 7.4 • Arterial PO2 = 100 mm Hg • Mixed venous PO2 = 40 mm Hg
Changes with Exercise • Temperature rises • pH drops • PO2 at the tissues decreases • PCO2 rises • No change in 2,3-DPG