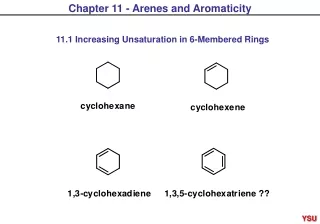
Arenes
Arenes. Introduction. Hydrocarbon that contain rings which stabilised by electron delocalisation are called aromatic hydrocarbon or arenes The simplest yet most important member of the aromatic hydrocarbons is benzene. Naming Aromatic Hydrocarbon.

Arenes
E N D
Presentation Transcript
Introduction • Hydrocarbon that contain rings which stabilised by electron delocalisation are called aromatic hydrocarbon or arenes • The simplest yet most important member of the aromatic hydrocarbons is benzene
Naming Aromatic Hydrocarbon • Monosubstituted alkylbenzene is named as derivatives of benzene. For example:
The IUPAC system retains certain common names for some simpler alkylbenzenes. For example: Aniline, styrene, toluene. • When two or more substituents on a benzene ring, three structural isomers are possible. • Location of two substituents may be indicated by the prefixsortho, meta or para • 1,2 is equivalent to ortho, o- • 1,3 is equivalent to meta, m- • 1,4 is equivalent to para, p- • The order of decreasing priorities of common substituents is: COOH > SO3H> CHO> CN > C=O> OH> NH2> R> NO2> X Where R= alkyl group ; X= halogen
When three or more groups are on the benzene ring, a numbering system must be used to locate them. For example,
In more complex molecules, the benzene ring is named as a substituent on a parent chain. In this case, the C6H5- group is called a phenyl group. For example
Exercise • Name the following aromatic compounds
Reaction of benzene • Nitration • Nitration is the substitution of a nitro group, -NO2, for one of the hydrogen atoms on the benzene ring • Benzene reacts with a mixture of concentrated nitric acid and concentrated sulphuric acid • The product is nitrobenzene, a pale yellow liquid
Halogenation • This reaction involves substitution of a halogen atom for a hydrogen atom on the aromatic ring • In the presence of a catalyst or halogen carrier such as aluminium chloride, AlCl3, benzene is substituted by chlorine or bromine at room temperature.
The catalyst aluminium chloride, AlCl3, an electron-deficient compound which has only six electrons in its outer shell, accepts a lone pair of electrons from one of the chlorine atoms, which induces polarisation in the chlorine molecule. • The positively charged end of the chlorine molecule is now an electrophile and attacks the benzene ring.
Alkylation: Friedel-Crafts reaction • This reaction substitutes an alkyl group for a hydrogen atom on an aromatic ring • When benzene is warmed with chloromethane, CH3Cl and aluminium chloride (catalyst) under anhydrous conditions, a substitution reaction occurs and methylbenzene is formed
AlCl3 polarise the CH3 Cl molecule • The positively charged methyl group attacks the benzene ring and electrophilic substitution occurs
Acylation • Involves substitution of an acyl group, RCO, for a hydrogen atom on the aromatic ring. • When benzene is added to ethanoyl chloride and AlCl3, under anhydrous conditions, a substitution reaction occurs and phenylethanone is formed.
Exercise • Write the structural formula for the producta formed from Friedal Crafts alkylation or acylation of benzene with (a) CH3CH2CH2Cl (b) CH3CH2CH2COCl
Sulphonation • Involves substitution of an –SO3H group for a hydrogen atom on the aromatic ring. • If benzene and [H2SO4] are reflux together for several hours, benzenesulphonic acid is formed:
Reaction of methylbenzene • Reaction with chlorine • When chlorine, Cl2, is bubbled through boiling methylbenzene in strong sunlight or uv light, substitution takes place on the methyl group:
If Cl2 is bubbled through methylbenzene in the absence of uv light but in the presence of AlCl3, the ring is substitutes. A mixture of two isomers is obtained. • White fumes of HCl are given off • Electron density of the benzene ring more strongly at the 2- and 4- positions
Reaction with HNO3 • When methyl benzene is added to HNO3 and concentrated H2SO4, at room temperature, a mixture of 2- and 4-nitromethylbenzene
If the temperature is raised, two or three nitro groups, NO2 are introduced
Reaction with CH3I (Friedal craft alkylation) • When methlbenzene is added to CH3I in AlCl3 at 20 oC, a mixture of 1,2-dimethylbenzene and 1,4-dimethylbenzene is formed
Reaction with CH3COCl (FriedalCraft Acylation) • The reaction of an acyl chloride with an aromatic hydrocarbon is known as FriedalCraft acylation. • Involves substitution of an acyl group, RCO-, for a hydrogen atom on the aromatic ring in the presence of AlCl3.
Exercise • Write the structural formula for the products formed by Friedal Crafts acylation or alkylation of methylbenzene with • C6H5COCl (b) CH3CH2Br
Reaction with SO3 • When methylbenzene is added to fuming H2SO4 containing SO3 at 0oC, a mixture of 2- and 4-methylbenzenesulphonic is obtained.
Oxidation of alkylbenzene • Alkylbenzenes are oxidised to benzoic acid on prolonged reflux with KMnO4 or K2Cr2O7
Exercise • Predict the products from treatment of each of the following compounds with KMnO4 / H2SO4 under reflux. (c) (a) (b)
Exercise • Name compound P, Q, R and S below
Exercise • Draw the structural formulae of the organic products formed when methylbenzene reacts with • Concentrated nitric acid and concentrated sulphuric acid at room temp • Chlorine in the presence of anhydrous aluminium chloride • Chloromethane in the presence of aluminium chloride








![The Synthesis of Sulfo Derivatives of Calix[4]arenes](https://cdn1.slideserve.com/3415659/slide1-dt.jpg)