Sodium Channel Structure, Function, Gating and Involvement in Disease
420 likes | 2.53k Views
Sodium Channel Structure, Function, Gating and Involvement in Disease. David R. Marks, M.Sc. . An Overview. Sodium Channel Structure - Current theory and Types of Na+ Channels Sodium Channel Function Current theory of inactivation Modulation Pharmacology Activation. An Overview Cont’d.

Sodium Channel Structure, Function, Gating and Involvement in Disease
E N D
Presentation Transcript
Sodium Channel Structure, Function, Gating and Involvement in Disease David R. Marks, M.Sc.
An Overview • Sodium Channel Structure - Current theory and Types of Na+ Channels • Sodium Channel Function • Current theory of inactivation • Modulation • Pharmacology • Activation
An Overview Cont’d • Article 2 – Na+ Channel Gating • Article 1 - Na+ Channels and Neurodegenerative Disease • Article 3 – Na+ channel mutation and physiology
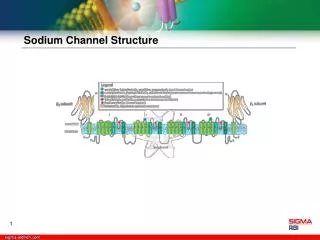
Sodium Channels - Structure • Composed of α, β-1 and β-2 subunits, but the large α-subunits carries most of the functional properties • 4 repeated motifs, each with 6 transmembrane domains • All linked together • Contain a voltage “sensor”/ligand binding domain (method of activation) • The hydrophobic S4 segment (voltage “sensor”) is found in all voltage gated Na+ channels and is absent in ligand gated Na+ channels • Selectivity filter (shell of hydration) • Inactivation gate
Cartoon representation of the “typical” voltage-activated sodium channel
Types Of Na+ Channels • Voltage gated – Changes in membrane polarity open the channel • Ligand gated (nicotinic acetylcholine receptor) – Ligand binding alters channel/receptor conformation and opens the pore • Mechanically gated (stretch receptor) – Physical torsion or deformation opens the channel pore
Sodium Channels - Function • Play a central role in the transmission of action potentials along a nerve • Can be in different functional states (3) -A resting state when it can respond to a depolarizing voltage changes -Activated, when it allows flow of Na+ ions through the -Inactivated, when subjected to a “suprathreshold” potential, the channel will not open
The theory is that the inactivation gate “swings” shut, turning off the channel
Please Keep In Mind • The structure of the Na+ channel is not 100% solved, hence a “working model” is drawn based on biophysical, pharmacological, physiological and molecular assays • Zhao (2004) writes “The mechanism of opening and closing is unknown, but structural studies suggest…”
Na+ Channel Modulation • Phosphorylation • sodium channel function is modulated by serine/threonine and tyrosine kinases as well as tyrosine phosphatases (Yu et al, Science 1997) • Mutation – altered amino acid sequence/structure can change the biophysical properties of the Na+ channel • Pharmacology – block Na+ channel to reduce the conductance • Proteolysis- (cleavage) Proteases may cleave specific residues or sequences that inactivate a channel, or significantly alter the biophysical properties
Why Na+ Channels/Modulation Are Important • Neuronal depolarization, Action Potential • Neuronal Excitability • Cardiac Excitability • Muscle Excitability • The basis of neuronal/cardiac/muscular function relies on the propagation of action potentials, down axons, sarcolemma, myocardium, as well as requiring synaptic transmission. • Differential excitability alters the electrical conduction/transmission properties of the “circuit”
Na + Channel Blockers/Pharmacological Agents • Tetrodotoxin (TTX) • Amioderone • Lidocaine • Procainamide • Mexilitine • Ketamine • Many, many others
Some Na+ Channels Outside The Nervous System • Naf – “Funny Current” in pacemaker cells of the heart (SA node/ectopic pacemakers) • Nav in the myocardium, sarcolemma, and T-tubules and motor endplate
Na+ Channel Activation • Change in transmembrane potential results in a conformation change in the Na+ channel • The four S4 segment alpha helices translocate, thus leading to the opening of the channel pore • The energy of the conformational change in the channel during activation is mediated by the reduction in overall entropy of the system. • The voltage sensor is a highly charged sequence of amino acids that “aligns” itself according to the electrical field present • A change in transmembrane potential creates unfavorable electrodynamic interaction for the voltage sensor, hence a conformational shift lowers the energy of the system and creates more favorable conditions
Patch Clamping/Transfection Transfection 1. Kv1.3 cDNA in Plasmid 2. Lipofectamine complexing 3. Add to Dishes 4. Patch 28-48 hrs after
Transition: A General Overview of Articles Before Discussion • From Basic structure/function relationships to a gating mechanism • The gating of a bacterial Na+ channel and application of Na+ channel activation and biophysical properties • Article 1 – A gating hinge in Na+ channels: a molecular switch for electrical signaling
Conserved glycine In the S6 domain Proposed conformational shift of A-helix caused by substitution of Proline for G219 Prolines in alpha helices after the first turn (4th residue) cause a kink in the helix. This kink is caused by proline being unable to complete the H-bonding chain of the helix and steric or rotamer effects that keep proline from adapting the prefered helical geometry
Na+ Channel Gating • Current theory holds that a change in transmembrane potential “flips” the conformation of the voltage sensor, thereby opening the channel pore • A mutation, G219P, glycine 219 changed to proline alters the conformation of the S6 domain • The mutant channel now favors a state much like the “open” state of a wild-type channel • NOTE: these bacterial Na+ channels are homotetramers of identical subunits
The G219P mutant activates significantly earlier (activates at much more negative voltages) than the wild-type V ½ : Voltage at which ½ of channels present are in the open state Comparable to Km in that it is a measure of the ability of a channel to activate
Other mutations to the Na+ channel Do not exert as significant effects in the activation (V ½)
Influence of hybrid Na+ channel subunits on gating and biophysical properties
Article 2 - Na+ Channels And Neurodegenerative Disease • Overview – Multiple Sclerosis (MS) displays a remission-relapse course. Some axons are able to maintain minimal conduction velocity, while others degenerate completely. • Definition: Experimental autoimmune encephalomytis (EAE) – animal model of MS
MS can display remission-relapsing course. This is believed to be the result of the expression of two distinct isoforms of voltage-gated Na+ channels NaV 1.2/1.6 are expressed over long distances (> 10μm)
B-amyloid are pepties associated with neurodegenerative diseases, and can accumulate in fibrillar aggregates
What is Important About This Article • Nav 1.6 is colocalized with a Na/Ca exchanger • Nav 1.2 is NOT colocalized with B-amyloid proteins • Nav 1.2 help restore conduction in demyelinated axons • Nav 1.6 is seen in degenerating axons
An increase in NaV1.6 yields an Increase in Na/Ca exchangers, elevating intracellular Ca2+ to harmful levels
Article 3 - Na+ Channels and the Conduction System of the Heart • Long QT syndrome – disease where the entire cycle of excitation-contraction coupling of the myocardium is prolonged • Patient had G-A substitution at codon 1763 of the Nav 1.5 channel gene, which changed a valine (GTG) to a methionine (ATG) • This mutation produced a persistently active and fast recovering Na+ channel • Mutant was INSENSITIVE to lidocaine
Article 3 • Authors generated a similar mutant by site-directed mutagenesis • Examined the mutant in a heterologous expression system to obtain biophysical and other properties
The Nav 1.5 V1763M mutant is Sensitive to TTX, but resistant to lidocaine TTX eliminates lidocaine-insensitive current
Why this is important: Other than traumatic cardiac arrest, arrhythmias degenerate into ventricular fibrillation or ventricular tachycardias. “circus movement” whereby tissue becomes “hyper-excitable”
Extension and Application of Na+ Channel Properties and Function Relating to Article 3
Advanced Cardiac Life Support(ACLS) Targets Na+ Channels Extensively • “Please Shock Shock Shock, Everybody Shock, And Lets Make Patients Better” The purpose of defibrillation of ventricular arrhythmias is to apply a controlled electrical shock to the heart, which leads to depolarization of the entire electrical conduction system of the heart. When the heart repolarizes, the normal electrical conduction may restore itself Depolarization theoretically inactivates all voltage-gated Na+ channels, and allows Voltage-gated potassium channels to activate, and help hyperpolarize the membrane
+40 mv -70 mv
V FIB/V TACH After phosphorylation/ phosphate cleavage After Administration Of Procainamide
Use-dependent block of sodium channels. • Blocks potassium channels. • Blocks alpha-adrenergic receptors. • Blocks muscarinic receptors. • Used to attempt to terminate persistent reentrant arrhythmias • Reduces automaticity of ALL pacemakers (both the SA node and ANY tissue capable of generating a pacemaker potential) • Slows Down Conduction of depolarization in ALL tissues of the heart and decreases cardiac excitability • This is your last resort. Giving this drug may stop the arrhythmia, but make it almost impossible for the heart to spread impulses after
Summary For the Lecture • Na+ channels are comprised subunits, the Alpha of 4 repeating motifs, each motif with 6 transmembrane domains • There are voltage, ligand, and mechanically-gated Na + channels • Na+ channels are involved in the depolarization of excitable membranes • Na+ channels have multiple modalities of modulation, which can alter neuronal/membrane excitability • Na+ channels are the target of a multitude of pharmacological agents
Summary • Na+ channels Are involved in the remission-relapse of MS • Na+ channel gating can be significantly affected by modulation (phosphorylation, mutation, proteolytic cleavage) • Mutation in Nav 1.5 is implicated in Long QT syndrome, generating persistent and slow inactivating sodium current