Batch Distillation
Batch Distillation. Pharmaceutical API Process Development and Design. Module Structure. Vapor Liquid Equilibrium Curves Rayleigh Distillation Column Configurations Column Operation Simulation Design of Batch Columns. Distillation. Used for separating a mixture of two or more liquids

Batch Distillation
E N D
Presentation Transcript
Batch Distillation Pharmaceutical API Process Development and Design
Module Structure Vapor Liquid Equilibrium Curves Rayleigh Distillation Column Configurations Column Operation Simulation Design of Batch Columns
Distillation • Used for separating a mixture of two or more liquids • Takes advantage of the differences in volatilities (vapor pressure) • For a binary mixture, αij – relative volatility, Pi0 – vapor pressure of pure liquid i
1 0 1 VLE Curve and BP/DP Curves T Saturated Vapor y Saturated Liquid 0 1 xA xA Mixture of A and B
If the mixture has a minimum-boiling azeotrope Homogeneous Azeotropes For non-ideal mixtures, the activity coefficients are different from unity: Phase diagrams for Isopropyl ether – Isopropyl Alcohol
If the mixture has a maximum-boiling azeotrope Homogeneous Azeotropes For non-ideal mixtures, the activity coefficients are different from unity: Phase diagrams for Acetone – Chloroform
For a minimum-boiling azeotrope with large deviation from Raoult’s law ( ), phase splitting may occur and a minimum-boiling heterogeneous azeotrope forms, having a vapor phase in equilibrium with two liquid phases. Heterogeneous Azeotropes • Homogeneous Azeotrope • Heterogeneous Azeotrope
Important properties of pure components, mixtures Vapor liquid equilibria Y-X diagrams, T-X, T-Y diagrams Existence of multiple liquid phases Commercial packages Part of process simulators Activity++, PPDS etc Helps you identify distillation boundaries Thermo Properties Calculations
Rayleigh Distillation Vapor Liquid Charge Heat L’, xi – remaining liquid and mole fraction at any subsequent time L’0, xi0 – initial liquid amount and mole fraction
For binary mixture when ij is constant Rayleigh Distillation (Contd)
Batch Evaporation Qc Accum 1 Accum 2 Qr
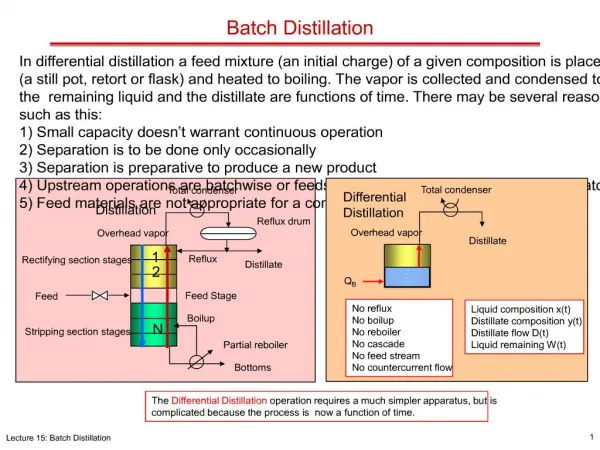
Preferred method for separation when Feed quantities are small Feed composition varies widely Product purity specification change with time High purity streams are required Product tracking is important Feed has solids Batch Distillation
Advantages Flexible Accurate implementation of recipe specific to a given mixture Several components separated using one column Requires least amount of capital Batch Distillation Advantages
Qc L D 1 • • Accum 1 Accum n N Qr Conventional Batch Distillation Column
Column Configurations Inverted BD Qc F F Qr Qr Accum 1 Accum n
Column Configurations Middle Vessel BD Qc Qc Accum 1 Accum n F F Qr Qr Accum n+1 Accum m
Side stream from the main column fed to a second column Can be used for mixtures with 3 or more components Take advantage of the build up of medium volatile component in the column Eliminate slop cut Reduce cycle time, energy consumption 260 Q2 262 A 217 2 266 270 216 Side Column 3 Main Column 218 219 222 220 B 1 214 Q3 232 223 224 228 C 230 240 Q1 Dual Column Configuration
Start-up period Vapor boilup rate policy Constant vapor boilup rate Constant condenser vapor load Constant distillate rate Constant reboiler duty Product period: Reflux ratio policy Shutdown period Column Operation
Operate under total reflux until the column reaches steady state (L / V = 1, R = ) Change reflux ratio to the desired value Collect distillate in accumulator End the ‘cut’ when certain criteria are satisfied Duration Condenser composition Accumulator composition, amount Reboiler composition, amount Column Operation Qc L D 1 • • Accum 1 Accum n N Qr
Increasing reflux ratio Improves separation Increases cycle time Increases energy consumption Profile optimization Trade-off between cycle time and value of recovered material Maximize profit Effect of Reflux Ratio
Staged Separation Qc V1 – vapor rate leaving plate 1 V L D 1 L / V – Internal reflux ratio L / D – Reflux ratio Vj, yj Lj-1, xj-1 Mj, xj N Plate j Lj, xj Vj+1, yj+1 Qr
Packed Columns • HETP – Height equivalent to one theoretical plate • Characteristic of packing • Number of plates = packed bed height/HETP
Simulation of Batch Distillation • Simulation of startup period • Simulation of product period • Column model • Examples • Benzene–toluene • Benzene–toluene–ortho-xylene • Acetone–chloroform
Dynamics of column during start-up are very difficult to model Rigorous model of tray hydraulics Rigorous model of heating column internals Typical simulation of start-up period Run column under total reflux until column reaches steady state At the beginning, assume that liquid compositions on plates and in the condenser are same as feed composition Simulation of Start-up Period
Total condenser without sub-cooling Perfect mixing of liquid and vapor on plates Negligible heat losses Condenser material balance Simulation of Product Period
Mass balance equations on plate j Column Model • Constant molar holdup • Constant volume holdup • VLE on each plate • Constraint
Enthalpy balance equations on plate j Column Model (Contd) • Physical properties
Vapor boilup rate from plate 1 is constant Quasi steady-state approximation During a small time interval, plate temperature, K values, vapor and liquid flowrates remain constant Solve the set of ODEs numerically up to the next update interval After each update interval, recompute bubble point, K values, plate enthalpies Vapor compositions Reboiler composition from mass balance Liquid and vapor flowrates from enthalpy derivatives Solution of Dynamic Model
Equimolar mixture of Benzene and Toluene 8000 liters charge Vapor boilup rate 20 kmol/hr Number of plates = 20 Plate holdup 4 liters Condenser holdup 180 liters Recover 99% mole fr Benzene and Toluene Simulated using BDIST-SimOpt Uses Activity++ physical properties package Benzene–Toluene Distillation
Benzene–Toluene–O-Xylene 20 plates
Acetone–Chloroform Azeotropic system
Use of Simulation in Batch Distillation • Synthesis of operating recipe and rapid characterization of batch distillations • Accurate determination of operating and design parameters of a batch column • Use in column operation to determine cut amounts and switching policy for each batch
Role of Simulation in Column Operation • Components • Cut Sequence • For each cut: • Starting and stopping criteria • Reflux ratio Simulator Model Developer Verified Model Simulator DCS Operator Column Feed Amount Feed Composition
Problems Related to Batch Distillation • Design of a batch column • Operating policy determination for individual column batches • Design and operation issues are interdependent
Design of Batch Columns • Main design parameters • Number of stages • Vapor boilup rate • Diameter • Still capacity (batch size) • Reboiler and condenser size heat transfer areas • Single separation duty • Multiple separation duties