Methods
Biology Core Curriculum, University of Wisconsin-Madison. D. Chen, R. Hommen, M. Kjetland , L . Loegering , M. Sunny. Results. Abstract. Discussion.

Methods
E N D
Presentation Transcript
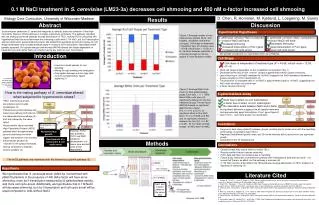
Biology Core Curriculum, University of Wisconsin-Madison D. Chen, R. Hommen, M. Kjetland, L. Loegering, M. Sunny Results Abstract Discussion Sacchromycescerevisiae (S. cerevisiae) responds to osmotic stress via activation of the High-Osmolarity Glycerol (HOG) pathway to increase intercellular osmolarity. This pathway interferes with the mating pheromone pathway through deactivation of TEC1, leaving FUS3untouched. We hypothesized that this would decrease the shmooing in cells with 0.1 M NaCl, but fus1 transcription and cell cycle arrest would remain the same. We tested this hypothesis by light microscopy for cell shape observation and a β-galactosidase assay to measure fus1 transcription. Hypotheses were partially accepted. Chi-square analysis and two-way ANOVA reveals cell shape dependent on treatment type and neither NaCl nor α-factor having any effect on fus1 transcription. Experimental Hypotheses Figure 1: Average number of cells observed per sample (N=8) with budded, unbudded, and shmoocell types. Cell shape is dependent of treatment type (chi-square value = 45.68, critical value = 12.59, df = 6). Variation between replicates is consistent enough for the chi-square to have statistical validity. Cell shape is independent of treatment type (Χ2 = 45.68, critical value = 12.59, df = 6) S. cerevisiae +α-factor/-NaCl(compared to -α-factor/-NaCl) will have: increased shmooing increased transcription of Fus1gene increased cell cycle arrest S. cerevisiae+α-factor/+NaCl (compared to +α-factor/-NaCl) will have: decreased shmooing equal transcription of Fus1gene equal cell cycle arrest • We partially accept both our hypotheses with low confidence. Introduction Cell Shape 0.1 M NaCl treatment in S. cerevisiae (LM23-3a) decreases cell shmooing and 400 nMα-factor increased cell shmooing • Used as a model species for cell signaling • Mating through budding and conjugation • Conjugation between a and α type cells involves complementary ligand • signaling (1) • Yeast cell shape is dependent on the 4 treatment combinations (Fig.1) • Decreased shmooing in both -α-factor groups suggests that α-factor causes shmooing. • Less shmooing in +α/+NaCl compared to +α/-NaCl suggests that NaCl decreases filamentous growth, possibly by TEC1 deactivation (3) • The proportion of unbudded cells in -α/+NaClis approximately equal to -α/-NaCl, suggesting that NaCl does not affect cell cycle arrest • α-factor causes shmooing S. cerevisiaewith bud scars, colors modified (http://www.kurzweilai.net/images/ Yeast.jpg) Figure 2: Average Miller Units (N=4) for beta-galactosidase assay. Error bars = +/- 1 SEM. Mean Miller units were not significantly different for the four treatment groups The two-factor ANOVA showed no significant effect for the α-factor (F(1,1)=1.14, p=0.31), no significant effect for the NaCl factor (F(1,1)=0.0046, p=0.94), and no significant interaction between the two (F(1,1)=1.82, p=0.20). All error bars overlap except –α/-NaCl and +α/+NaCl. How is the mating pathway of S. cerevisiae altered when subjected to hyperosmotic stress? β-galactosidase assay • TEC1 (filamentous growth transcription factor) builds cytoskeleton (3) • Filamentous growth pathway and fusion pore pathway both start as the same pheromone pathway (2); both are induced by the same ligand • Hyperosmotic stress activates High-Osmolarity Glycerol (HOG) pathway which phosphorylates tyrosine and threonine which triggers transcription of an enzyme that produces glycerol (3) • Glycerol in the cytosol increases internal osmolarityto maintain osmotic gradient (4) • No significant difference suggests that all sample treatments • had approximately equal transcription of fus1 gene (figure 2) • Type II error – incorrectly accept null hypotheses NaClhas no effect on fus1 transcription α-factor has no effect on fus1 transcription No interaction exists between NaCl and α-factor Confidence Deactivation of tec1 • Large error bars show variability between groups, possibly due to human error with the specificity of the assay, could lead to type II error • Replicates had different growth times, but 30 minute intervals did not account for any significant growth • Assumptions met for statistical test However, FUS3 also activates FUS1 and FAR1 leading to development of a fusion pore and cell cycle arrest, respectively No filamentous growth Development of a fusion pore & cell cycle arrest Methods Conclusions • Osmotic stress may induce HOG1 to inhibit TEC1 • Results confirm α-factor induces shmooing • FUS1 data with NaCl inconclusive due to variability • Future study: interaction of pheromone pathway with methylglyoxal acid and citric acid – it is known that there is an effect, but the pathway is unknown (6) • 0.1 M NaCl osmotic stress affects the HOG pathway causing deactivation of TEC1 leading to a decrease in shmooing (4) The HOG pathway only interferes with the filamentous growth pathway (2). Hypothesis We hypothesize that S. cerevisiaestrainLM23-3a, transformed with pBH315 plasmid, in the presence of 400 nMα-factor will have more shmooing, more fus1 transcription measured by β-galactosidase activity, and more cell cycle arrest. Additionally, we hypothesize that 0.1 M NaCl will decrease shmooing, but fus1 transcription and cell cycle arrest will be equal compared to cells without NaCl. Literature Cited 1. HoopesB., Pruitt N. L., Baier K., Brooks S. 1998. Signal transduction and control of the cell cycle in the yeast (Saccharomyces cerevisiae). Biocore Lab Manual 2013:58-69MclClean et all 2007 2. Shock T.R., Thompson J., Yates J.R., Madhani. 2009. Hog1 mitogen-activated protein kinase (MAPK) interrupts signal transduction between the Kss1 MAPK and the Tec1 transcription factor to maintain pathway specificity. Eukaryotic Cell 8(4): 606-616 3. O’Rourke S.M., Herskowitz I., O’Shea E.K. 2002. Yeast go the whole HOG for the hyperosmotic response. Trends in Genetics 18(8):405-412 4. Nolan S., Cowan A.E., Koppel D.E., Jin H., Grote E. 2006. FUS1 Regulates the opening and expansion of fusion pores between mating yeast. Molecular Biology of the Cell 17(5): 2439-2450. 5. O’Rourke S.M., Herskowitz I., O’Shea E.K. 2002. Yeast go the whole HOG for the hyperosmotic response. Trends in Genetics 18(8):405-412 6. Hernandez-Lopez, M., Randez-Fil, F., and Prieto, J. 2006. Hog1 mitogen-activated protein kinase plays conserved and sitinct roles in the osmotolerant yeast. Eukaryotic Cell 5(8): 5574-5582. NaCl and α-factor added at the same time Hoopeset al 1998