Mineral Formation
Mineral Formation . 6 th Grade. Popcorn Rock Activity. When the rock dolomite is doused in white distilled vinegar, aragonite crystals slowly form. A similar process occurs in Yellowstone National Park and in the Carlsbad Caverns in New Mexico. Carlsbad Caverns, New Mexico.

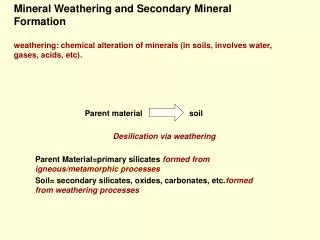
Mineral Formation
E N D
Presentation Transcript
Mineral Formation 6th Grade
Popcorn Rock Activity • When the rock dolomite is doused in white distilled vinegar, aragonite crystals slowly form
A similar process occurs in Yellowstone National Park and in the Carlsbad Caverns in New Mexico
Stala…WHAT?! StalagMITE = on the ground –mighty and tall StalacTITE= hanging from the ceiling –hanging tight to the ceiling
These form similarly to the “popcorn rock” • Acid rain or water from other sources reacts with the rock such as limestone or dolomite • This reaction hardens into a mineral • “Popcorn rocks” appear on cavern walls, such as at the Lehman Caves
Crystals that often form from water added to rock are usually calciteand aragonite
What are 3 ways that minerals form? • 1) form by magma and lava cooling • 2) form by hot water solutions • solutions: elements and compounds that are mixed with water • 3) form by solutions evaporating
Geode = a rounded, hollow rock that is often lined with mineral crystals
Formed when…. • 1) a cavity forms in a rock –(an open space, similar to a shell) –usually happen with rocks that are made from lava (the empty space is the gas bubbles)
Geode –crystals form when water containing dissolved minerals seeps into a crack or hollow in a rock
Minerals from Solutions • Solution: a mixture in which one substance is dissolved in another
1) Minerals formed from Evaporation • Minerals form when solutions evaporate • Ex. If you stir salt crystals into a beaker or water, the salt dissolves, forming a solution. Eventually the solution evaporates, leaving behind salt crystals • Ex. mineral halite formed over millions of years when ancient seas slowly evaporated • This happens in the US along the Midwest, Southwest, and along the Gulf Coast
2) Minerals from Hot Water Solutions • Deep underground, magma heats water to a high temperature • When the water solution begins to cool, the elements and compounds leave the solution and crystalize as minerals • Often PURE metals are formed this way
Minerals from Hot Water Solutions • Vein = a narrow channel or slab or a mineral that is different from the surrounding rock
Remember that PURE METALS tend to form from hot water solutions underground, forming veins • Ex. Gold, Silver, Platinum
Platinum • Luster: metallic • Fracture: hackly • Hardness: 4.5 • Streak: silver-grey • Cleavage: none
Silver • Luster: metallic • Fracture: hackly • Hardness: 2.5 – 3 • Streak: silver-white • Cleavage: none
3) Minerals formed from magma and lava: • Minerals form: • As hot magma cools inside the crust • As lava hardens on the surface *When liquids cool to a solid state, they form crystals. Tourmaline: a mineral that forms as magma cools deep beneath the surface What influences the size of crystals?
The size of crystals depend on: • The rate at which magma cools • The amount of gas the magma contains • The chemical composition of the magma Magma closer to the surface cools FASTER (smaller crystals) Magma deep within the crust cools SLOWLY (bigger crystals)
The size of a mineral depends on whether its high or low in the crust • Large crystals –deep within the crust • Small crystals --near the surface
http://www.youtube.com/watch?v=0OLdSJmvcUs • Take a look!