Introduction to Gas Chromatography
480 likes | 943 Views
Introduction to Gas Chromatography. Written by Bette Kreuz Produced by Ruth Dusenbery University of Michigan-Dearborn 2000. Introduction. Gas chromatography is an instrumental method for the separation and identification of chemical compounds. Slide 1a. A sample is

Introduction to Gas Chromatography
E N D
Presentation Transcript
Introduction to Gas Chromatography Written by Bette Kreuz Produced by Ruth Dusenbery University of Michigan-Dearborn 2000
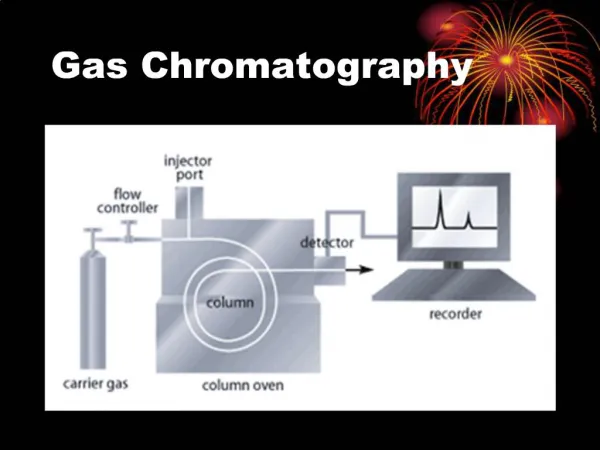
Introduction • Gas chromatography is an instrumental method for the separation and identification of chemical compounds.
Slide 1a • A sample is • introduced into a heated injector, • carried through a separating column by an inert gas, and • detectedas a series of peaks on a recorder when components leave the column.
Slide 2a • The instrument used for our experiments, a Varian 3350 gas chromatograph, is shown here.
Slide 2 Computer Controls for Method and Output Carrier gas/ Regulator Varian 3350 Gas Chromatograph
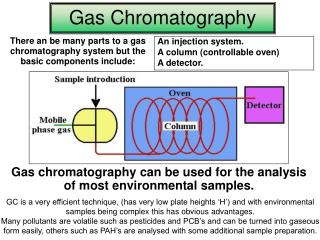
Slide 3a • Chromatographic separation involves the use of a stationary phase and a mobile phase. • Components of a mixture carried in the mobile phase are differentially attracted to the stationary phase and thus move through the stationary phase at different rates.
Slide 3 Flow of Mobile Phase Injector Detector T=0 T=10’ T=20’ Most Interaction with Stationary Phase Least
Seperation • Separation of the components of the mixture occurs in the column. • Compounds differentially retained in the stationary phase reach the detector at different times to produce a set of peaks along the time line.
Slide 4a • In gas chromatography • the mobile phase is an inert carrier gas and • the stationary phase is a solid or a liquid coated on a solid contained in a coiled column.
Slide 5a • Columns can be short, large diameter packed column or long, very small diameter capillary columns. • Each has its own use and associated advantages and disadvantages.
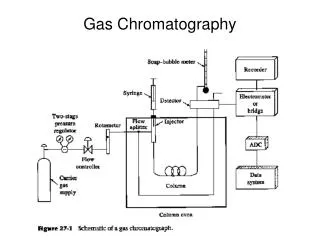
Slide 6a • The mobile phase or carrier gas flows through the instrument from a pressurized tank. • Flow rate is controlled by a two stage regulator on the gas tank and additional controls within the instrument.
Slide 6 Two Stage Tank Regulator GC Flow Controller
Slide 7a • The column is contained in a heated oven that is preceded by a heated injector port and followed by a heated detector unit which produces the output. • A set of preprogrammed parameters regulate the operation of the system.
Slide 8a • The injector, column oven and detector components of the Varian 3350 gas chromatograph are shown here.
Slide 8 Injector Detector Column in Oven
Slide 9a • Preprogrammed parameters, called a separationmethod, control the operation of the system.
Slide 9 Control Panel sets Separation Method
Slide 10a • Samples may be pure compounds. • However, they are often prepared as dilute solutions due to the sensitivity of the detection methods.
Slide 10 Dilute Solution Pure Sample
Slide 11a • When the system is ready, as indicated by the ready light, samples are injected into the injector port where they are vaporized and carried into the column by the carrier gas.
Slide 11 10 ml Syringe
Exit to Detector Enter from Injector Slide 12 Packed Column installed in Oven Compartment.
Slide 13a • The detector response is sent to a computer system where the progress of the sample is monitored on the computer monitor in graphical form that displays detector response as a function of run time.
Slide 14a • Each component of the mixture reaches the detector at a different time and produces a signal at a characteristic time called a retention time. • The area under a peak is related to the amount of that component present in the mixture.
Slide 15a • The detector information can also sent to a printer that produces hard copy of the chromatographic run.
Slide 16a In the printout of the chromatographic analysis: the number of peaks correlates with the number of components in the sample, the area under each peak correlates with the relative amount of that component in the sample, and if standard information is available, the retention time under defined conditions can be used to identify each component.