ATP
Making energy!. ATP. The point is to make ATP !. Chemical energy. (a). First law of thermodynamics: Energy can be transferred or transformed but Neither created nor destroyed. For example, the chemical (potential) energy in food will be converted to the kinetic

ATP
E N D
Presentation Transcript
Making energy! ATP The pointis to makeATP!
Chemical energy (a) First law of thermodynamics: Energy can be transferred or transformed but Neither created nor destroyed. For example, the chemical (potential) energy in food will be converted to the kinetic energy of the cheetah’s movement in (b). Figure 8.3 First Law Of Thermodynamics
Heat co2 + H2O (b) Second law of thermodynamics: Every energy transfer or transformation increases the disorder (entropy) of the universe. For example, disorder is added to the cheetah’s surroundings in the form of heat and the small molecules that are the by-products of metabolism. Figure 8.3 Second Law
∆G < 0 ∆G = 0 Reactions in a Closed System: What would happen To a living System if it were closed?
∆G < 0 Body Cells: What do we Need to Stay alive?
The energy needs of life • Organisms are endergonic systems • What do we need energy for? • synthesis • building biomolecules • reproduction • movement • active transport • temperature regulation
Where do we get the energy from? • Work of life is done by energy coupling • use exergonic (catabolic) reactions to fuel endergonic (anabolic) reactions digestion energy + + synthesis energy + +
Living economy • Fueling the body’s economy • eat high energy organic molecules • food = carbohydrates, lipids, proteins, nucleic acids • break them down • digest=catabolism • capture released energy in a form the cell can use • Need an energy currency • a way to pass energy around • need a short term energy storage molecule ATP Whoa!Hot stuff!
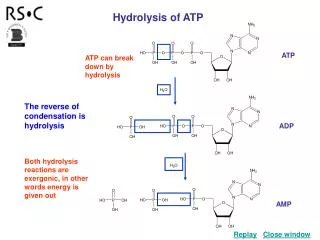
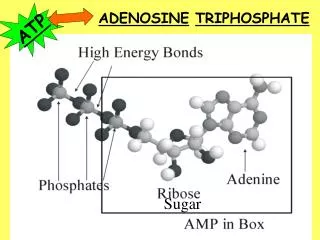
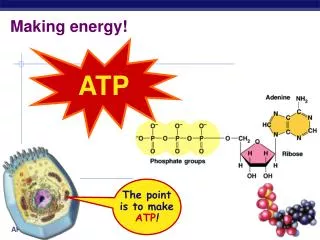
high energy bonds ATP • Adenosine TriPhosphate • modified nucleotide • nucleotide =adenine + ribose + Pi AMP • AMP + Pi ADP • ADP + Pi ATP • adding phosphates is endergonic How efficient! Build once,use many ways
O– O– O– O– O– O– O– O– P P P P P P P P –O –O –O O– O– O– –O –O –O O– O– O– –O –O O– O– O O O O O O O O I thinkhe’s a bitunstable…don’t you? How does ATP store energy? • Each negative PO4 more difficult to add • a lot of stored energy in each bond • most energy stored in 3rd Pi • 3rd Pi is hardest group to keep bonded to molecule • Bonding of negative Pi groups is unstable • spring-loaded • Pi groups “pop” off easily & release energy ADP AMP ATP Instability of its P bonds makes ATP an excellent energy donor
O– O– O– O– P P P P –O O– –O O– –O –O O– O– O O O O How does ATP transfer energy? • ATP ADP • releases energy • ∆G = -7.3 kcal/mole • Fuel other reactions • Phosphorylation • released Pi can transfer to other molecules • destabilizing the other molecules • enzyme that phosphorylates = “kinase” 7.3energy + ADP ATP
enzyme + + H H H H H H H H H2O C C C C C C C C OH OH HO HO O O + H ATP ADP + C H H P HO OH H C C + + Pi C P An example of Phosphorylation… • Building polymers from monomers • need to destabilize the monomers • phosphorylate! synthesis +4.2 kcal/mol “kinase”enzyme It’snever thatsimple! -7.3 kcal/mol -3.1 kcal/mol
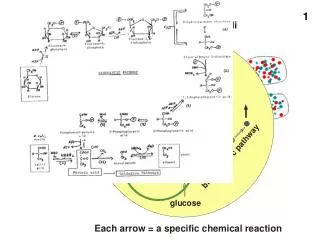
P ATP C 2 hexokinase C 2 ADP phosphofructokinase H C P Another example of Phosphorylation… • The first steps of cellular respiration • beginning the breakdown of glucose to make ATP glucose C-C-C-C-C-C Thosephosphatessure make ituncomfortablearound here! fructose-1,6bP P-C-C-C-C-C-C-P DHAP P-C-C-C G3P C-C-C-P activationenergy
+ Pi ATP / ADP cycle ATP • Can’t store ATP • good energy donor, not good energy storage • too reactive • transfers Pi too easily • only short term energy storage • carbohydrates & fats are long term energy storage cellularrespiration 7.3 kcal/mole ADP A working muscle recycles over 10 million ATPs per second Whoa!Pass methe glucose (and O2)!
Cells spend a lot of time making ATP! Thepoint is to makeATP! What’s thepoint?
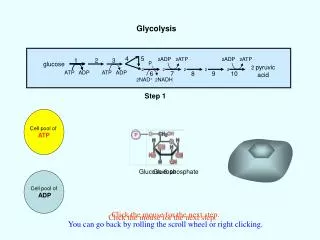
How is ATP Made in a Cell? Substrate Level Phosphorylation
Chemiosmosis Start with a mitochondrion or chloroplast Trap H+ in the intermembrane space
Chemiosmosis Start with a mitochondrion or chloroplast Trap H+ in the intermembrane space How can this lead to ATP production?
H+ H+ H+ H+ H+ H+ H+ H+ rotor rod + P catalytic head H+ ATP synthase • Enzyme channel in mitochondrial membrane • permeable to H+ • H+ flow down concentration gradient • flow like water over water wheel • flowing H+ cause change in shape of ATP synthase enzyme • powers bonding of Pi to ADP:ADP + Pi ATP ADP ATP But… How is the proton (H+) gradient formed?
That’s the rest of mystory! Any Questions?