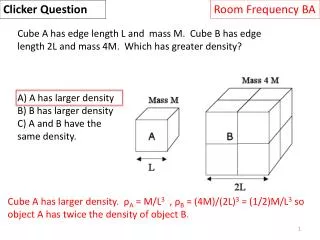
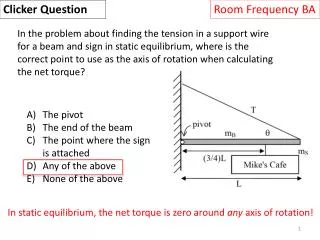
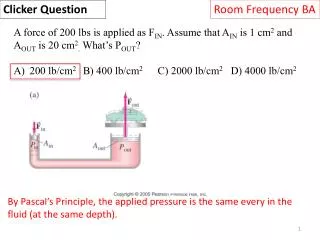
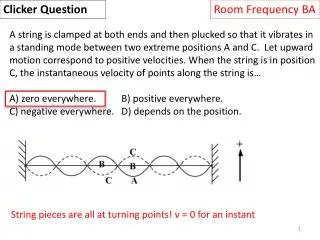
Clicker Question
Clicker Question. Which of the following quantities is the largest? A. 2.0 km B. 2.0 cm C. 2.0 mm D. 2.0 Gm. Example 2a . (Clicker Question). The number of significant figures in 400000 m is: A. 1 B. 2 C. 4 D. 6. Example 2b . (Clicker Question).

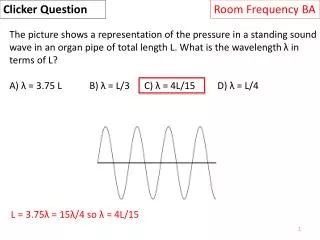
Clicker Question
E N D
Presentation Transcript
Clicker Question • Which of the following quantities is the largest? • A. 2.0 km • B. 2.0 cm • C. 2.0 mm • D. 2.0 Gm
Example 2a. (Clicker Question) • The number of significant figures in • 400000 mis: • A. 1 • B. 2 • C. 4 • D. 6
Example 2b. (Clicker Question) • The number of significant figures in 200.0 mL is: • A. 4 • B. 3 • C. 2 • D. 1
Example 2c. (Clicker Question) • The number of significant figures in • 0.000105000 gis: • A. 2 • B. 3 • C. 6 • D. 9 • E. 10
Example 3a. (Clicker Question) • 4378.99 to 3 SF is: • A. 4.38 • B. 438 • C. 4380 • D. 4380. • E. 4380.00
Example 3b. (Clicker Question) • 0.004588 to 2 SF is: • A. 0.0046 • B. 0.004600 • C. 46 • D. 4600 • E. 4.6 x 10-2
Example 3c. (Clicker Question) • 3900000 to 4 SF • A. 3.900 • B. 3900 • C. 3900. • D. 3.900 x 106 • E. 3.900 x 107
Example 3d. (Clicker Question) • 10 to 5 SF • A. 1.0000 • B. 10.000 • C. 10000. • D. 10.000 x 10-5 • E. 10.000 x 105
Example 5b. (Clicker Question) • 164 lb in kilograms (kg) is: • A. 0.164 • B.74.4 • C. 360.8 • D. 2.624x103
Example 5c. (Clicker Question) • 700.0 μL in mL is: • A. 7.000 x 10-7mL • B. 7.000 x 10-5mL • C. 0.7000 mL • D. 7.000 x 105mL
Clicker Question • Sulfur will most likely __ electrons to become stable. • A.gain two • B.gain six • C. lose two • D. lose six
Clicker Question • Magnesium will prefer to __ electrons and will have a __ charge. • A. Gain two; -2 • B. Gain two; +2 • C. Lose two; -2 • D. Lose two; +2
Example 2. (Clicker Question) • The chemical formula formed by magnesium ion andchloride ion is: • A. MgCl2 • B. Mg2Cl • C.MgC2 • D. Mg2C
Example 2. (Clicker Question) • The chemical formula formed by iron (III) ion andchloride ionis: • A. FeCl3 • B. Fe3Cl • C. IrCl3 • D.Ir3Cl
Example 3b. (Clicker Question) • The name of Ba3N2 is: • A. Barium nitride • B.Barium(III) nitride • C.Barium(II) nitride • D. Barium nitrogen • E. Tribariumdinitrogen
Example 4c. (Clicker Question) • The charge on the transition metal is __. • The name of the compound is __. • A. +2; vanadium(II) sulfide • B. +2; vanadium(II) sulfur • C. +4; vanadium(IV) sulfide • D. +4; vanadium(IV) sulfur
Example 4d. (Clicker Question) • The charge on the transition metal is __. • The name of the compound is __. • A. +1; magnesium(I) fluoride • B.+1; manganese(I) fluoride • C. +7; magnesium(VII) fluoride • D. +7; manganese(VII) fluoride
Example 6. (Clicker Question) • The name of PCl5 is: • A. Monophosphorus pentachloride • B. Monopotassium pentachloride • C. Phosphorus pentachloride • D. Potassium pentachloride
Example 7. (Clicker Question) • The chemical formula formed by iron (III) ion and ClO3- is: • A. FeClO4 • B. FeClO7 • C. FeCl3O12 • D. Fe(ClO4)3
Example 7. (Clicker Question) • The chemical formula formed by sodium ion and sulfate ion is: • A. NaSO4 • B. Na2SO4 • C. Na(SO4)2 • D. Na2(SO4)2 • E. NaS2O4
Example 7. (Clicker Question) • The chemical formula formed by iron (II) ion and sulfate ion is: • A. FeSO4 • B. FeS2O4 • C. Fe2(SO4)2 • D. Fe2(SO4)3 • E. Fe3(SO4)3
Example 7. (Clicker Question) • The chemical formula formed by iron (III) ion and sulfate ion is: • A. FeSO4 • B. Fe2(SO4)2 • C. Fe2(SO4)3 • D. Fe3(SO4)3 • E. Fe3S3O4
Example 8b. (Clicker Question) • The name of Mg(ClO4)2 is: • A. Magnesium perchlorate • B. Magnesium (II) perchlorate • C. Manganese perchlorate • D. Manganese (II) perchlorate
Example 8c. (Clicker Question) • The name of Fe(OH)3 is: • A. Iron hydroxide • B. Iron (I) hydroxide • C. Iron (III) hydroxide • D. Iron trihydroxide
Example 8d. (Clicker Question) • The name of NH4Cl is: • A. Ammonium chlorate • B. Ammonium chloride • C. Nitrogen perchlorate • D. Nitrogen tetrahydrogenchloride
Example 9c. (Clicker Question) • The formula mass for MgSO4 is: • A. 72.38 amu • B. 120.38 amu • C. 151.01 amu • D. 184.38 amu
Example 9c. (Clicker Question) • The formula mass of lithium sulfate is: • A. 39.01 amu • B. 103.01 amu • C. 109.95 amu • D. 199.08 amu
Example 10b. (Clicker Question) • The number of moles in 4.32 x 1022 molecules of aspirin is: • A. 0.0718 • B. 13.9 • C. 7.18 x 1044 • D. 7.18 x 1046
Example 11b. (Clicker Question) • The no. of moles present in 12.25 g of aspirin (C9H8O4) is: • A.0.04861 mol • B.0.06798 mol • C.14.71 mol • D.2207 mol
Example 1b. • What is the electron geometry around C (a), C (b) and C (c)? • A. tetrahedral; tetrahedral; tetrahedral • B. tetrahedral; trigonal planar; tetrahedral • C. tetrahedral; linear; pyramidal • D. tetrahedral; bent; trigonal planar
Example 1b. • What is the molecular shape around C (a), C (b) and C (c)? • A. tetrahedral; tetrahedral; tetrahedral • B. tetrahedral; trigonal planar; tetrahedral • C. tetrahedral; linear; pyramidal • D. tetrahedral; bent; trigonal planar
Example 1c. • What is the electron geometry around C (a) and C (b) ? • A. linear; linear • B. tetrahedral; tetrahedral • C. tetrahedral; linear • D. bent; bent
Example 1c. • What is the molecular shape around C (a) and C (b) ? • A. linear; linear • B. tetrahedral; tetrahedral • C. tetrahedral; linear • D. bent; bent
Example 1a. (Clicker Question) • 102.3 °F in °C is: • A. 24.8 °C • B. 39.1 °C • C. 216 °C • D. 242 °C
Example 1b. (Clicker Question) • 27.5 °C in °F is: • A. 47.3 °F • B. 49.5 °F • C. 81.5 °F • D. 107 °F
Example 2b. (Clicker Question) • _____ has a higher boiling point, because _____. • A. PH2Cl, it has dipole forces • B. NH2Cl, it has H-bonding • C. PH2Cl, it has a higher molecular weight • D. NH2Cl, it has dipole forces
Example 2c. (Clicker Question) • _____ has a higher boiling point, because _____. • A. GeBr4, it has dipole forces • B. GeH4, it has H-bonding • C. GeBr4, it has a higher molecular weight • D. GeH4, it has dipole forces
Example 4. (Clicker Question) • 425 mmHg in atm and bar is ____ and ____, respectively. • A. 0.559 atm; 0.566 bar • B. 0.559 atm; 0.552 bar • C. 1.79 atm; 1.81 bar • D. 3.23x105atm; 3.27x105 bar
Clicker Question • A cylinder with a movable piston has a volume of 150 mL. If the temperature is kept constant, while the pressure inside the cylinder is doubled, the volume of the cylinder: • A. Will also be doubled • B. Will be halved • C. Will be squared • D. Will not change
Clicker Question • A balloon has a volume of 150 mL. If the pressure is kept constant, while the temperature is halved, the volume of the balloon: • A. Will also be doubled • B. Will be halved • C. Will be squared • D. Will not change
Example 13. (Clicker Question) • Where do you expect your soda to have more bubbles, at the beach or in the mountains? • A. At the beach; pressure is higher, hence more dissolved CO2 • B. In the mountains; pressure is higher, hence more dissolved CO2 • C. At the beach; pressure is lower, hence more dissolved CO2 • D. In the mountains; pressure is lower, hence more dissolved CO2
Example 3b. (Clicker Question) • 150.0μg/mL in g/L is: • A. 1.5 x 10-4g/L • B. 1.500 x 10-4g/L • C. 0.015 g/L • D. 0.15 g/L • E. 0.1500 g/L
Example 2b. (Clicker Question) • The molecules in 2b are: • A. Different bec. they have different chemical formulas • B. Different bec. they are structural isomers • C. Identical
Example 2c. (Clicker Question) • The molecules in 2care: • A. Different bec. they have different chemical formulas • B. Different bec. they are structural isomers • C. Identical
Example 2d. (Clicker Question) • The molecules in 2dare: • A. Different bec. they have different chemical formulas • B. Different bec. they are structural isomers • C. Identical
Example 4c. (Clicker Question) • The molecular formula of4cis: • A. C3H6O2 • B. C3H6O3 • C. CH3CH(OH)CHO • D.
Example 4d. (Clicker Question) • The expanded structural formula of4dis: • A. B. • C. D.
Example 4e. (Clicker Question) • The expanded structural formula of4eis: • A. B. • C. D.
Example 5c. (Clicker Question) • The molecule in5c: • A. YES geom. isomerism; CIS • B. YES geom. isomerism; TRANS • C. NO geom. isomerism; CIS • D. NO geom. isomerism; TRANS • E. NO geom. isomerism; no cis/trans
Clicker Question • What will be thecondensed structure of butane? • A. C. • B. D.