CITRIC ACID CYCLE -Pyruvate Dehydrogenase
CITRIC ACID CYCLE -Pyruvate Dehydrogenase Reading: Harper’s Biochemistry Chapter 18 Lehninger Principles of Biochemistry 3rd Ed. pp. 567-583 OBJECTIVES

CITRIC ACID CYCLE -Pyruvate Dehydrogenase
E N D
Presentation Transcript
CITRIC ACID CYCLE-Pyruvate Dehydrogenase Reading: • Harper’s Biochemistry Chapter 18 • Lehninger Principles of Biochemistry 3rd Ed. pp. 567-583
OBJECTIVES • To understand how the pyruvate dehydrogenase complex acts as a mediator of flow of carbon compounds between the glycolytic pathway and the citric acid cycle. • To understand the central role of the citric acid cycle in mitochondrial energy metabolism. • The structures of the intermediates in the cycle. • The names of the enzymes that catalyze each step. • The cofactors and products involved in each step.
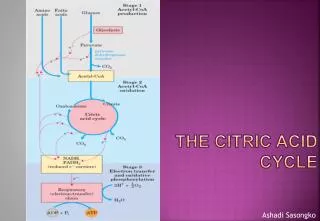
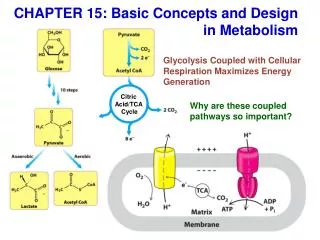
The citric acid cycle (Krebs cycle, tricarboxylic acid cycle) is a series of reactions in mitochondria that bring about the catabolism of acetyl residues. • The acetyl groups are fed into the citric acid cycle, which enzymatically oxidizes them to CO2. The energy released by oxidation is conserved in the reduced electron carriers NADH and FADH2. • The reduced coenzymes are then oxidized themselves, giving up protons and electrons. The electrons are transferred, eventually, to O2, via a chain of electron-carrying molecules known as the respiratory chain. • In the course of electron transfer, the large amount of energy released is conserved in the form of ATP by a process termed oxidative phosphorylation.
Catabolism of proteins, fats, and carbohydrates in the three steps of cellular respiration
Biomedical Importance • The citric acid cycle acts as a final common pathway for the oxidation of carbohydrate, lipids, and protein, because glucose, fatty acids, and many amino acids can all be metabolized to acetyl-CoA or intermediates in the cycle. • The citric acid cycle also plays a major role in gluconeogenesis, transamination, deamination, and lipogenesis. The liver is the only tissue where all of these occur to a significant extent. • When large numbers of liver cells are damaged or destroyed, in acute hepatitis or cirrhosis, this can have major repercussions on metabolism. • Very few genetic abnormalities exist for enzymes of the citric acid cycle, suggesting such abnormalities are incompatible with normal development and highlighting the vital nature of the process. • Pyruvate hydrogenase is a key enzyme needed to convert pyruvate to acetyl-CoA. A variety of disorders in pyruvate metabolism are due to defects in this enzyme.
Pyruvate transport into mitochondria • Pyruvate generated in the cytoplasm by glycolysis must be transported across the inner mitochondrial membrane via a pyruvate/H+ symport. • This transport uses some of the energy stored in the mitochondrial inner membrane electrical potential gradient.
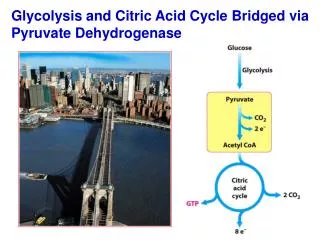
Conversion of pyruvate to acetyl-CoA by pyruvate dehydrogenase • Within the mitochondria, pyruvate is oxidatively decarboxylated to acetyl-CoA. • This reaction is catalyzed by several different enzymes working sequentially in a multi-enzyme complex collectively designated as the pyruvate dehydrogenase complex. • Overall reaction: • Or Simply: Pyruvate + NAD+ + CoAAcetyl-CoA + NADH + H+ + CO2
Coenzyme A (CoA) • Coenzyme A (CoA). A hydroxyl group of pantothenic acid is joined to a modified ADP moiety by a phosphate ester bond, and its carboxyl group is attached to -mercaptoethylamine in amide linkage. The hydroxyl group at the 3´position of the ADP moiety has a phosphoryl group not present in ADP itself. The -SH group of the mercaptoethylamine moiety forms a thioester with acetate in acetyl-coenzyme A (acetyl-CoA)
Pyruvate Dehydrogenase Complex • Contains three enzymes each present in multiple copies: 1. pyruvate dehydrogenase (E1) 2. dihydrolipoyl transacetylase (E2) 3. dihydrolipoyl dehydrogenase (E3) • The complex from mammals has 60 copies of E2, which contains lipoate, and constitutes the core of the complex. • In addition, five different coenzymes or prosthetic groups are needed: 1. thiamine pyrophosphate (TPP) 2. flavin adenine dinucleotide (FAD) 3. coenzyme A (CoA) 4. nicotinamide adenine dinucleotide (NAD) 5. lipoate • All are clustered for efficient handling of intermediates
Lipoic acid (lipoate) in amide linkage with the side chain of a Lys residue. The lipoyllysyl moiety is the prosthetic group of dihydrolipoyl transacetylase (E2 of the pyruvate dehydrogenase complex). The lipoyl group occurs in oxidized (disulfide) and reduced (dithiol) forms and acts as a carrier of both hydrogen and an acetyl (or other acyl) group.
Oxidation decarboxylation of pyruvate to acetyl-CoA by the pyruvate dehydrogenase complex
Pyruvate reacts with thiamine pyrophosphate bound to E1. Pyruvate undergoes decarboxylation to the hydroxylethyl derivative, with a loss of CO2. • The acetyl group and 2 electrons from TPP are transferred to the oxidized form of the lipoyllysyl group of the core enzyme (E2). • A transestenification occurs where the -SH group of CoA replaces the -SH group of E2 to yield acetyl-CoA and the reduced form of the lipoyl group. • E3 transfers two hydrogens from the reduced lipoyl groups to E2 to FAD. • The reduced FADH2 of E3 transfers a hydride ion to NAD+, forming NADH.
Biomedical Implications • Mutations in the genes for the subunits of the pyruvate dehydrogenase complex, or a dietary thiamine deficiency, can have severe consequences. • Thiamine-deficient animals are unable to oxidize pyruvate normally. Particularly important in the brain, which usually obtains energy from the aerobic oxidation of glucose. e.g. Beriberi is a disease that results from thiamine deficiency and is characterized by loss of neural function. • Alcoholics can also develop thiamine deficiency because much of their dietary intake of calories is vitamin-free. • An elevated level of blood pyruvate is often an indicator of defects in pyruvate oxidation.
Overview of citric acid cycle • Eight successive reaction steps. • The six carbon citrate is formed from two carbon acetyl-CoA and four carbon oxaloacetate. • Oxidation of citrate yields CO2 and regenerates oxaloacetate, which plays essentially a catalytic role. • The energy released is captured in the reduced coenzymes NADH and FADH2.
Step 1: Formation of citrate • Citrate synthase catalyzes the condensation of acetyl-CoA with oxaloacetate to form citrate • Oxaloacetate binds first and induces a conformational change, creating a binding site for acetyl-CoA • CoA is liberated and recycled
Step 2: Formation of isocitrate via cis-aconitate • The enzyme aconitase catalyzes the reversible transformation of citrate to isocitrate, through the intermediary formation of the tricarboxylic acid cis-aconitate. • The reaction proceeds to the right because isocitrate is rapidly consumed in the next step of the cycle.
Step 3: Oxidation of isocitrate to -ketoglutarate and CO2 • Isocitrate dehydrogenase catalyzes oxidative decarboxylation of isocitrate to form -ketoglutarate
Step 4: Oxidation of -ketoglutarate to succinyl-CoA and CO2 • This step is another oxidative decarboxylation, in which -ketoglutarate is converted to succinyl-CoA and CO2 by the -ketoglutarate dehydrogenase complex. The energy of oxidation of the substrate is conserved in the formation of the thioester bond of succinyl-CoA • This reaction is virtually identical to the pyruvate dehydrogenase reaction, except that E1 of the pyruvate dehydrogenase complex binds pyruvate, and E2 of the -ketoglutarate dehydrogenase complex binds -ketoglutarate • Arsenite inhibits the reaction causing -ketoglutarate to accumulate.
Step 5: Conversion of succinyl-CoA to succinate • In this step, the thioester bond of the substrate is broken and used to drive the synthesis of a phosphoanhydride bond in either GTP or ATP, forming succinate. • The enzyme becomes phosphorylated at a His residue as an intermediate in this reaction, and this is transferred to ADP or GDP to form ATP or GTP. Animal cells have two isozymes, one specific for ADP and one for GDP. If GTP is formed, ATP can be generated by nucleoside diphosphate kinase: GTP + ADP GDP + ATP
Step 6: Oxidation of succinate to fumarate • The flavoprotein succinate dehydrogenase oxidizes succinate to fumarate • In eukaryotes, succinate dehydrogenase is tightly bound to the inner mitochondrial membrane, and is the only enzyme of the citric acid cycle that is membrane-bound. • Malonate, an analog of succinate, is a strong competitive inhibitor of succinate dehydrogenase and therefore blocks the citric acid cycle.
Step 7: Hydration of fumarate to malate • The reversible hydration of fumarate to L-malate is catalyzed by fumarase (fumarate hydratase)
Step 8: Oxidation of malate to oxaloacetate • In the last reaction of the citric acid cycle, NAD-linked L-malate dehydrogenase catalyzes the oxidation of L-malate to oxaloacetate • Under standard thermodynamic conditions, the equilibrium of this reaction lies far to the left, but in cells, oxaloacetate is continually removed by the highly exergonic citrate synthase reaction in step 1. This keeps the concentration of oxaloacetate in the cells low (10-6M).
Products of one turn of the citric acid cycle. Three NADH, one FADH2, one GTP (or ATP), and two CO2 are released in oxidative decarboxylation reactions. All cycle reactions are shown in one direction only, but keep in mind that most of the reactions are reversible. The two carbons appearing as CO2 are not the same two carbons that entered the cycle as the acetyl group of acetyl-CoA (they are in oxaloacetate, and will be released in subsequent cycles) The cycle generates the equivalent of 12 ATP’s from one acetyl-CoA (3 NADH = 9 ATPs, 1 FADH2 = 2 ATPs, 1 ATP (GTP) directly)
Vitamins play key roles in the citric acid cycle • Four of the soluble vitamins of the B complex have precise roles in the functioning of the citric acid cycle: 1. Riboflavin - in the form of flavin adenine dinucleotide (FAD), a cofactor in the dehydrogenase complexes. 2. Niacin - in the form of nicotinamide adenine dinucleotide (NAD), a cofactor for three dehydrogenases. 3. Thiamine/vitamin B - as thiamine dephosphate, the coenzyme for the -ketoglutarate dehydrogenase reaction. 4. Pantothenic acid - a part of coenzyme A, the cofactor attached in acetyl-CoA and succinyl-CoA