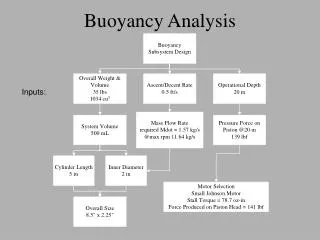
Buoyancy
Buoyancy. Archimedes Principle.

Buoyancy
E N D
Presentation Transcript
Archimedes Principle • Archimedes' Principle: When an object is partially or fully submerged, the buoyant force, or the apparent loss in weight, is equal to the weight of the fluid displaced. Buoyancy: Buoyancy is a force exerted on an object in a fluid environment (this includes air). This results in the object going up and opposing gravity. • Affect: An object may have positive buoyancy, if the pressure below the object is larger then the pressure above the object, then object will rise. • Apparent weight: The apparent weight equals the actual weight minus the buoyant force. • Affect: In order to have buoyancy an object must have a lower average density then the fluid it is in.
Avogadro’s Principle • Avogadro's law: Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules. • Affect: Two tanks of equal volume at the same temperature and pressure contain the same number of molecules regardless of what gas they are. • However: In order for it to be convenient, chemists have chosen one temperature and one pressure as a standard, chemists have chosen 1 atm to be the standard pressure and 0°C to be standard pressure temperature. These conditions are called STP (Standard Temperature and Pressure) • 1 mole (6.02 ×10 molecules) of any gas at STP occupies 22.4 L
Charles’s Law • Charles's Law: At a constant pressure, the volume of any sample of gas increases as its temperature increases, and vice versa. • Affect: Because a balloon can stretch, the pressure inside is constant. When the air in the balloon is heated, its volume increases, expanding the balloon. Now since the balloon has expanded, the size has increased but not the amount of air in the balloon. In other words, the volume of air in the balloon has increased, but the mass has remained the same. Since the air in the balloon has becomes less dense then the air outside the balloon, the air outside provides a Buoyant force on the balloon and gives the balloon lift.