Ultraviolet Light
160 likes | 708 Views
Ultraviolet Light. Vanderbilt Student Volunteers for Science Spring 2019. IA. Introduction - What is the Electromagnetic Spectrum?. Give each student: one electromagnetic spectrum handout. One instruction sheet. Explain the electromagnetic spectrum: Light is an electromagnetic wave.

Ultraviolet Light
E N D
Presentation Transcript
Ultraviolet Light Vanderbilt Student Volunteers for Science Spring 2019
IA. Introduction - What is the Electromagnetic Spectrum? • Give each student: • one electromagnetic spectrum handout. • One instruction sheet. • Explain the electromagnetic spectrum: • Light is an electromagnetic wave. • In physics, light refers to electromagnetic radiation of any wavelength. • Different types of waves are named according to wavelengths, frequencies and energies – radio, microwave, infrared, visible, ultraviolet, x-rays, & gamma. • Visible light is made of many colors. • Explain that the focus is on ultraviolet light • Mention that the sun emits the whole EM spectrum, but the atmosphere is able to block out most harmful parts of the spectrum. • Fun mnemonic device for the spectrum: Raging Martians Invaded Venus Using X-ray Guns
IB. Introduction - What is UV Light? • UV stands for ultraviolet, a portion of the light spectrum beyond what our eyes can see. • Explain that UV light has a shorter wavelength and thus has a higher energy than that of visible light. • Draw a wave on the board and show the students how a wavelength is measured.
IC. Introduction - What Happens When UV Light Reaches the Earth’s Atmosphere? • Ask the students if they know how the earth’s atmosphere is protected from too much UV radiation. • Most UV radiation is absorbed by the ozone layer in the stratosphere or reflected back into space. In the upper atmosphere ozone is a “good” gas since it screens out dangerous UV rays. • Ozone information: • It is an essentially colorless gas (pale blue at high concentrations) that has a distinctively sharp and unpleasant smell. • Ozone is naturally formed in the atmosphere when UV rays react with O2. • Ozone hole information: • An ozone hole forms over Antarctica every spring. • Ozone depletion is primarily caused by chlorine contained in chlorofluoro carbons (CFC’s).
IIA. The Integumentary System • Ultraviolet light has more energy than visible light and can damage living cells. This can cause cancer, cataracts, sunburn… • Ask students if they know what the integumentary system is? • Skin, hair, and nails make up the integumentary system. • The skin is the body's largest organ. • Pass out the skin models – 1 per group of 4. • Ask students to give some important functions of skin. • Its purpose is to protect the body from damage, infection and drying out. • It has two main layers: the inner layer, called the dermis, and the outer layer, called the epidermis • Briefly discuss the functions of the 2 layers
IIB. How is UV Light Dangerous? • Most UV radiation is absorbed by the atmosphere or reflected off. • There are three categories of UV light: UVA, UVB, and UVC • UVA: (at 400-315nm) is the closest to the visible light. • UVB: (315-290nm) is mostly absorbed by the ozone layer in the atmosphere. • UVC: (290-220nm) is very dangerous, but it is all blocked by the earth’s atmosphere (the ozone layer) • UVA rays can age us and UVB rays can burn us • Refer back to the skin model to show how deep the UV rays can penetrate. • UVA rays penetrate deep into the dermis. • UVB rays will usually burn the superficial layers of your skin. It can also damage the eye tissue. • UV light damages many things other than human skin – see discussion on Prescription bottles and milk jugs.
IIB. What Are Some Ways To Block UV Light? • We should block as much UV light from our bodies as is possible. • What are some ways to block UV light? • Sunscreen, long sleeves and pants, wide brimmed hats, sunglasses, and staying in the shade. • Avoid tanning beds and tanning lotions.
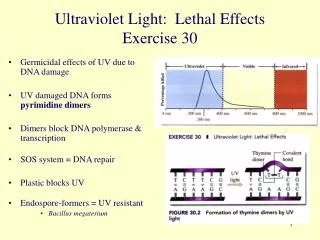
III. Testing UV Blocking Using UV Sensitive Beads • Tellstudents they will be testing several items to see how well they block UV radiation (all students and VSVSers wear goggles). • Divide the students into pairs and give each group a UV light. • Show the students a string of UV detecting beads. Point out that all the beads are white. • Expose the beads to a portable UV light until the beads have turned color. The beads contain a chemical that absorbs UV light and reemits it as visible light. The purple beads will be used in this experiment because they change to a more intense color. • Explain that the color stays for some time after the necklace is no longer exposed to UV radiation. This is an example of phosphorescence.
III. Testing UV Blocking Using UV Sensitive Beads (cont.) • Discuss the use of sunscreen to prevent sunburn. • Special chemicals absorb UV light, preventing it from reaching your skin. • How do you know how well a sunscreen blocks UV light? • All sunscreen is labeled with an SPF (Sun Protection Factor) number that indicates how well it absorbs UV light. • SPF 15 means it should take 15 times as long for skin damage to occur as it would on unprotected skin. • Who should wear sunscreen? • Everyone, even those with dark skin. UV exposure increases the risk for developing skin cancer.
III. Testing UV Blocking Using UV Sensitive Beads (cont). • Demonstrate procedure, steps 1-7 , then let students work in pairs. A control bead completely exposed to UV light, sunscreen, a sunglass lens, T-shirt material and medication bottle will be tested. 1. Put 3 beads in the squares on the first row of the observation sheet, and cover with the lens of a sunglass, and the piece of T-shirt. Leave one bead uncovered – this is the control bead. Tell the students they will be exposing these items to UV light and will be observing the color changes that occur. 2. Hold the UV light about 2-4 inches above the beads (make sure all beads have UV light shining on them at the same time). 3. When the control bead has turned purple, turn the UV light off and remove the lens and material. Observe and record the color of the beads.
III. Testing UV Blocking Using UV Sensitive Beads (cont.) 4. Place the remaining 3 beads in the squares on the 2nd row. • Place a medication bottle over one bead. • Show the students the acetate sheets, Q-tips and the containers of SPF 45 sunscreen. Dip a Q-tip in the sunscreen, spread a good amount on the acetate, and immediately put on top of the bead, sunscreen side UP. (Tell them this is important to prevent the sunscreen from drying out.) Emphasize that they should try to avoid getting any sunscreen on the paper and beads, since it is difficult to wash off. 7. Repeat the procedure in #1. Tell the students to carefully remove the acetate sheets covered with sunscreen and place them on the paper towel with the Q-tips. Keep all sunscreen away from lens etc. A VSVS member will collect all materials with sunscreen on them and put them in the marked bag. We will wash beads for reuse.
IV. Review results • Ask students: • How can you test if the goggles prevent UV light from damaging their eyes? By placing a UV detecting bead inside up-turned goggles and holding the goggles over the black light. • Have students perform this test if there is time. • Ask students: • Does sunscreen really work? Yes • Do sunglasses block UV? Yes • You may want to elaborate that these sunglasses were cheap. • Does clothing protect you from UV light? Yes
Clean up Clean-Up and Return of the Kit: • Students should place everything (except the acetate, paper towel and Q-tips used for sunscreen) back into their plastic bags. • A VSVS member will collect all materials with sunscreen on them and put them in the marked bag. • VSVS members will collect and return everything to the VSVS lab.
V. UV-Detecting Bracelets • Give each student a container of 5 UV beads and 1 piece of braid for string beads. • Tell the students to string the beads onto the braid. After they have finished stringing the beads, they should make a bracelet by putting the untied end through the hole in the bead on the tied-off end and tying a knot. • When they are finished, they should shine their black lights on the beads. • Students can keep the bracelets and measure the amount of UV radiation on sunny days. • Note: Be sure to collect all black lights. Count to make sure you have 16. Check that they are turned off and place them back in the kit box before you go on with the next sections.
VI. Optional. How Can UV Light Be Useful? • Ask students if they know any uses for UV lights. • Mention antimicrobial, insecticidal, and forensic uses of UV light. • Tell students to look at their handout sheet to see the $20 bill showing the fluorescing strip.