HETEROGENEOUS CATALYSIS AN INTRODUCTION
700 likes | 1.44k Views
HETEROGENEOUS CATALYSIS AN INTRODUCTION. Paul Ratnasamy National Chemical Laboratory Pune-411008, India. Why R& D in catalysis is important. - 27 % of GNP and 90 % of chemical industry involve products made using catalysts (food, fuels, polymers, textiles, pharma/agrochemicals,etc)

HETEROGENEOUS CATALYSIS AN INTRODUCTION
E N D
Presentation Transcript
HETEROGENEOUS CATALYSISAN INTRODUCTION Paul Ratnasamy National Chemical Laboratory Pune-411008, India
Why R& D in catalysis is important -27 % of GNP and 90 % of chemical industry involve products made using catalysts (food, fuels, polymers, textiles, pharma/agrochemicals,etc) -For discovery/use of alternate sources of energy/fuels/ raw material for chem industry. -For Pollution control-Global warming. - For preparation of new materials (organic & inorganic-eg: Carbon Nanotubes).
Catalysis is multidisciplinary (physics,chemistry & chem engg) • The catalyst is an inorganic solid;Catalysis is a surface phenomenon;solid state and surface structures play important roles. • Adsorption,desorption and reaction are subject to thermodynamic, transport and kinetic controls(chem engg); • adsorbate-substrate and adsorbate - adsorbate interactions are both electrostatic and chemical(physical chemistry). • The chemical reaction is organic chemistry.
Green Chemistry is Catalysis • Pollution control(air and waste streams; stationary and mobile) • Clean oxidation/halogenation processes using O2,H2O2(C2H4O, C3H6O, ECH) • Avoiding toxic chemicals in industry ( HF,COCl2 etc.) • Fuel cells( H2 generation)
Catalysis in Nanotechnology Methods of Catalyst preparation are most suited for the preparation of nanomaterials . • Nano dimensions of catalysts. • Common prep methods. • Common Characterization tools. • Catalysis in the preparation of carbon nanotubes.
Hetrogeneous Catalysis-Milestones in Evolution-1 • 1814- Kirchhoff-starch to sugar by acid. • 1817-Davy-coal gas(Pt,Pd selective but not Cu,Ag,Au,Fe) • 1820s –Faraday H2 + O2H2O(Pt);C2H4 and S • 1836- Berzelius coins”Catalysis”; • 1860-Deacon’s Process ;2HCl+0.5O2 H2O + Cl2; • 1875-Messel.SO2 SO3 (Pt); • 1880-Mond.CH4+H2O CO+3H2(Ni); • 1902-Ostwald-2NH3+2.5O22NO+3H2O(Pt); • 1902-Sabatier.C2H4+H2 C2H6(Ni). • 1905-Ipatieff.Clays for acid catalysed reactions; isomerisation, alkylation, polymerisation.
Milestones in Evolution-2 • 1910-20: NH3 synthesis (Haber,Mittasch) ; Langmuir • 1920-30-Methanol syn(ZnO-Cr2O3); Taylor;BET • 1930-Lang-Hinsh &Eley -Rideal models ;FTsyn;EO; • 1930-50:Process Engg; FCC / alkylates;acid-base catalysis;Reforming and Platforming. • 1950-70: Role of diffusion; Zeolites, Shape Selectivity; Bifunctional cata;oxdn cat-HDS; Syngas and H2 generation. • 1970- Surface Science approach to catalysis(Ertl) • 1990 - Assisted catalyst design using : -surface chem of metals/oxides, coordination chemistry - kinetics,catalytic reaction engg - novel materials(micro/mesoporous materials)
Catalysis in the Chemical Industry • Hydrogen Industry(coal,NH3,methanol, FT, hydrogenations/HDT,fuel cell). • Natural gas processing (SR,ATR,WGS,POX) • Petroleum refining (FCC, HDW,HDT,HCr,REF • Petrochemicals(monomers,bulk chemicals). • Fine Chem.(pharma, agrochem, fragrance, textile,coating,surfactants,laundry etc) • Environmental Catalysis(autoexhaust, deNOx, DOC)
PHYSICAL ADSORPTION • Steps in a catalytic Reaction: - Diffusion of reactant (bulk, Film, surface) - Adsorption( physical chemical) -Surface reaction - Desorption and diffusion of products • Physical Adsorption: - Van der Waals forces;BET surface area • Pore Size distribution ( Wheeler, de Boer, BJH) • Influence of pore size on reaction order, temperature coefficient, selectivity, Influence of poisons …
CHEMISORPTION • Langmuir isotherm; Langmuir –Hinshelwood and Eley- Rideal mechanisms of surface reactions;Kinetics of adsorption-Elovich equation. Uses of chemisorption (1)probes (H2,CO,NH3, pyridine,CO2) for fraction of catalytically active surface (only 0.1% in cracking);(2)Do chemisorbed species actually participate in reactions(isotope exchange);(3) changes in surface structures on adsorption(S, H2, O2, H2O2…).
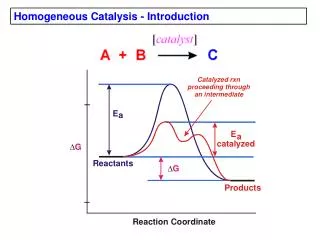
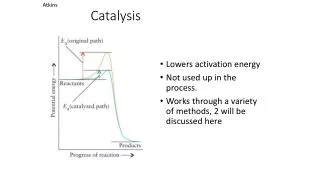
The Sabatier Principle “There is an optimum of the rate of a catalytic reaction as a function of the heat of adsorption”- Sabatier,1905: If the adsorption is too weak,the catalyst has little effect;If too strong, the adsorbates will be unable to desorb from the surface;Hence,the interaction between reactants or products with surface should be neither too strong nor too weak.
Sabatier Principle -Optimal basicity results in high carbonate yields (MMM 90(2006)314)
How catalysts accelerate rates of chemical reactions • H2+0.5O2 H2O; G 0298 = -58 Kcal/mol; In the gas phase: • D(H-H) = 103 and D(O-O)=117 Kcal/mol; • E# ~ 10 Kcal/mol for H+O2 or H2+O HO2 or H2O. Hence,kinetically gas-phase reaction improbable. Pt forms Pt-H and Pt-O bonds with E# ~ 0;Moreover, Pt-H + Pt-O Pt-OH Pt -OH2 has E# ~ 0 .
Turnover frequencies, Rates and numbers CATALYSIS IS A KINETIC PHENOMENON Sequence of elementary steps in steady state: diffusion (bulk,film,surface) - adsorption-reaction-desorption-diffusion TOF= number of product molecules formed per unit area per sec(molecules.cm-2.sec-1) TOF= number of product molecules formed per active site per sec(molecules.sec-1) onlyif active site is known. TOT= 1/TOF = turnover time, time necessary to form a product molecule(sec); TOR = Turnover rate = TOF X Surface area TON= TOF X total reaction time;TON=1( stoichiometry); TON must be >100 to be industrially useful.
Conversions,Rates and Rate constants • Conversion = % Reactant converted; • Reaction rate = kpX f(Pi) or kcX f(Ci) • k = Aexp(-E#/RT);A is temp independent. • TOFs between 0.0001 and 100 in industry; Temp adjusted to get the desired rates. • E# ~ 35-45 Kcal/mol for isom,cyclisation, cracking,dehydo/hydrogenolysis;HighT needed. • E# ~ 6-12 Kcal/mol for hydrogenation;
The Compensation Effect • k = A exp(-E#/RT); • For a given reaction, over different catalysts, A increases linearly with E# so that k remains constant: ln A = + (E# / R ); is a constant and is the isokinetic temp,when the rates on all catalysts are equal; A plot of ln A vs E# gives a linear plot with +ve slope.
Compensation effect for the methanation reactionLogarithm of preexponential factor vs apparent activation energy
The Active SiteH.S.Taylor,Proc Roy Soc (London)A108(1925)105 • “There will be all extremes between the case in which all the atoms in the surface are active and that in which relatively few are so active “. • “The amount of surface which is catalytically active is determined by the reaction catalyzed”.
Active Sites-Metals:Structure sensitivity of Catalytic reactions over metals • Structure Sensitive if rate changes markedly when crystallite/particle size is changed; “active site” comprises ensemble of many metal atoms;steps & edges. eg:hydrogenolysis,H2-D2 exch, steam reform,coking, aromatization etc • Structure Insensitive if rate is independent of crystallite /particle size; each surface metal atom is a potential active site; example: hydrogenation, dehydrogenation
Active Sites-Oxides /Sulfides.Catalysis by Ions at surfaces • Bronsted & Lewis acids in solution • Solid acid catalysts-Historical(acid-washed clays for cat cracking) • L acidity of ions:Na+< Ca 2+<Y3+<Th 4+.increases with charge/radius ratio. • B acidity by ion substitution (Al for Si) in clays, zeolites, Al phosphates etc. • Acidity measurement ( Total, L & B ).
Heterolytic adsorption on Ionicoxide surfaces Oxide Surface: M+-O - - M+ - O - - M+ Lewis acid(e- acceptor) Bronsted base(H acceptor) H+ H- H2: M+- O - - M+ - O - - M+ H+OH- H2O: M+ -O --M+- O - - M+ (B acid and B base) C2H5H H OCH3 C2H6 & CH3OH: M+ -O - - M+ - O - - M+
“Life Cycle” of a Catalyst • Catalyst Preparation • Activation • Surface reconstruction during catalytic run - Beneficial-Sulfiding of Re in PtRe - Harmful (carbon formation) • Deactivation( poisons,coke, SA loss, leaching) • Regeneration • Catalyst Unloading
Activity, Selectivity, Stability and Accessibility • High activity per unit volume . • High selectivity for desired product at adequate conversion level (STY for product > 1mol / ml/sec) • High Accessibility;Role of transport rates of mass and heat. • Long life time; Regenerability. • Thermal/mechanical strength in reaction conditions(sintering,crushing,attrition) • Reproducible/economic/safe manufacture.
CATALYST CHARACTERIZATION • Bulk Physical Properties • Bulk Chemical properties • Surface chemical properties • Surface Physical Properties • Catalytic Performance
Bulk Chemical Properties • Elemental composition( of the final catalyst ), EPMA • XRD,electron microscopy (SEM,TEM). • Thermal Analysis(DTA/TGA). • NMR/IR/UV-Vis/ EPR/ Mossbauer • TPR/TPO/TPD • EXAFS
Surface Properties • XPS,Auger, SIMS(bulk & surface structure). • Texture :Surface area- porosity. • Counting “Active” Sites: -Selective chemisorption (H2,CO,O2, NH3, Pyridine,CO2);Surface reaction (N2O). • Spectra of adsorbed species (IR/EPR/ NMR / EXAFS etc)
Physical properties of formulated catalysts • Bulk density • Crushing strength & attrition loss (comparative) • Particle size distribution • Porosimetry( micro(<2 nm),macro(>35 nm) and meso.
Catalyst Activity Testing :Definitions- Activity • Activity may be expressed as: -Rate constants or TON from kinetics -Rates/weight -Rates/volume -Conversions at constant P,T,and SV. - Temp required for a given conversion at constant partial & total pressures - Space velocity required for a given conversion at constant pressure and temp
Catalyst Activity TestingDefinitions- Selectivity • Selectivity = % concentration of product(s) among all the products excluding coke. • Yield = conversion X selectivity. • Selectivities may depend on T,P,SV,diffusion, catalyst particle size and shape , reactor geometry etc. • Always compare selectivities at constant T,P and most important,conversion. • Selectivity w.r.t. each of the reactants(H2O2).
Catalyst Testing- 1 • What is the objective ?Testing a solid for its catalytic properties in many reactions?screening for a particular reaction? Exploring Kinetics?Industrial development? • Activity;comparison at non-diffusion & non-thermodynamically limited, kinetically controlled conditions; • 10-20 mesh;dreactor >10diacat(wall effects) • Bed length/ dreactor >5 to avoid channeling; • Comparison of Selectivity at similar activity;
Catalyst Testing-2 Only at intermediate conversions and at low temp can the quality of the catalyst, expressed in an optimum of kinetically controlled conversion,be analyzed.At high temp or at high conversions,all catalysts are almost equal for either slow kinetic control or thermodynamically limited conversion.
Start-Up Procedures Affect Catalyst Performance Activated as per manfacturers instruction Activated Rapidly
Catalyst Preparation & Formulation -1 Catalyst Formulation - Size and shape is a compromise between the wish to minimize pore diffusion effects( small size)and pressure drop( large size); - Pelleting,extrusion,granulation,spray drying; Choice depends on properties of powder, size/shape/density/ required strength of catalyst particle; -Loading of graded sized pellets.
Catalyst Preparation & Formulation-2 • Unsupported Metals - very high activity(small area adequate ) - High purity feedstock eg: NH3 NO ( Pt-Rh gauze). CH3OH HCHO (Ag granules) - Raney Ni,Co,Cu for H2 ion (residual Al2O3 present!).
Catalyst Preparation & Formulation- 3 • Fused catalysts. eg: Triply promoted Fe ( + Ca,K,Al as oxides) catalyst for NH3 synthesis. Fe3O4 + H2(N2 +H2) Fe(1600C) Melt the mixture at 1600 C,cool,crush,size.
Catalyst Preparation & Formulation- 4 • Wet methods of catalyst manufacture: (A) Precipitation :pH of precipitating medium critical !! (B)Precipitation-deposition: texture of support important. Influence of Ageing,digestion; filterability; washability of salts;
The pH of precipitation affects chemical composition, particle size and other physical properties of Cu/ZnO/Al2O3 WGS shift catalyst
Catalyst Preparation & Formulation- 5 • Supported Metal(especially noble metals) Catalysts: • Used Extensively in industry: -autoexhaust, diesel oxidation, DeNOx, stationary power sources - Hydrocracking,Naptha reforming,xylene isom, isomerisations, Hydrogenations, etc - Fuel cell catalysts - Major issues: high cost and loss of activity due to sintering .
Why the need for high dispersion of PM • PM are expensive: hence impregnation and not coprecipitation • Activity depends on metal surface area (MSA) • MSA increases with dispersion
Metal Dispersion • Metal Dispersion, D = No of Pt surface atoms / No of Total Pt atoms D is an operational definition (defined by technique used) N total= from chemical composition N surface is obtained by physical or chemical methods Physical methods: Crystallite size from XRD, SEM/TEM Chemical methods: Chemisorption of H2, CO, H2-O2 titration PM distribution Profiles a.Uniform b.Egg shell c.Egg white d.Egg yolk
PM distribution profiles • Optimal dispersion depends on • reaction kinetics and mode of catalyst poisoning • Attrition strength of catalyst • Egg shell favors • Reactions with positive order • Fast reactions - Egg Yolk favors • Reactions with negative order • Pore mouth poisoning egg white or egg yolk • Low attrition strength egg white or egg yolk
Factors affecting dispersion of PM -1 • Concentration of PM • Low concentration – high dispersion • Presence of competing ions in impregnating solution increases D. • Citric acid in H2PtCl6 impregnation on Al2O3 platforming)
Factors influencing dispersion of PM -2 3.Functional groups on substrate surface for binding the PM precursor – Point of zero charge (PZC) influences dispersion of PM Anions and neutral complexes disperse better on gamma Al2O3 at pH<8 PZC gamma alumina=8-9; SiO2~3
Factors influencing dispersion of PM -3 4.Crystallite size of substrate Al2O3, CeO2, CZO, TiO2 etc Small crystallite sizes have large dispersion 5. Partially reducible oxide supports increaseD eg Pt-CeO2 6. Ion exchange of PM increases D, eg: Pt in zeolites
Sintering of PM • Leads to lower dispersion, MSA and activity • Increases with PM loading • Increases with T, TOS, H2O, O2, S, Cl • Increases with crystallite size of support • Increases with hydrophobicity of support (Pt-SiO2 sinters more than Pt-Al2O3) • Suppressed by spacers (ZrO2 in CZO) • Suppressed by “binding” groups on surface (OH, Cl-, SO3H- etc)
Reverse Micro Emulsion (RME) method enables use of lower amount of Pt in DeNOx • Nissan WO 2005/063391A1, PCT WO 2006/067912 A1 and others • Catalyst was first used in a Nissan engine using gasoline fuel for 30 hrs at 700ºC. • After engine durability test for 50 hrs at 70ºC, catalyst was tested in test rig at 350ºC for DeNOx activity. • Catalyst=100g/l in honeycomb; Pt-Co(Ce)-Al2O3 0.5% Pt is as effective as 3%wt Pt At 350ºC after endurance test at 700ºC for 30 hrs
Some Developments in Industrial catalysis-11900- 1920s Industrial ProcessCatalyst 1900s:CO + 3H2 CH4 + H2O Ni Vegetable Oil + H2 butter/margarine Ni 1910s:Coal Liquefaction Ni N2+3 H2 2NH3 Fe/K NH3 NO NO2 HNO3 Pt 1920s: CO +2 H2 CH3OH (HP) (ZnCr)oxide Fischer-Tropsch synthesis Co,Fe SO2 SO3 H2SO4 V2O5
Heterogeneous Catalysis.Some Challenges Ahead • Selective oxdn of long chain paraffins to terminal alcohols/ald/acids; • CH4CH3OH. • Activation of CO2 & its use as raw material; CO2 + H2O/ CH3OH/C2H5OH C2+ • Chiral catalysis with high ee. • H2 generation from H2O without using HC . • Photocatalysis with Sunlight.
Industrial catalysis-21930s and 1940s 1930s:Cat Cracking(fixed,Houdry) Mont.Clay C2H4C2H4O Ag C6H6 Maleic anhydride V2O5 1940s:Cat Cracking(fluid) amorph. SiAl alkylation (gasoline) HF/acid- clay Platforming(gasoline) Pt/Al2O3 C6H6 C6H12 Ni