Catalysis
Catalysis. Reaction Rate can be increased by increasing the concentration of reactants increasing the surface area of reactants increasing the temperature. Catalysis. Sometimes increasing the temperature to increase the reaction rate is not feasible

Catalysis
E N D
Presentation Transcript
Catalysis • Reaction Rate can be increased by • increasing the concentration of reactants • increasing the surface area of reactants • increasing the temperature
Catalysis • Sometimes increasing the temperature to increase the reaction rate is not feasible • in biological systems, high temperatures can destroy living cells • in a factory, high temperatures require a lot of energy, often at high cost
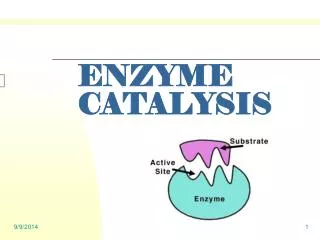
Catalysis • Enzymes • increase the rates of reactions in living cells • almost every biologically important reaction is assisted by a specific enzyme
Catalysis • Catalyst • a substance that speeds up a reaction without being consumed • an enzyme is a biological catalyst • catalysts are used in industry to speed up reactions
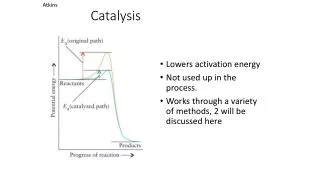
Catalysis • How does a catalyst work? • Lowers the activation energy of a reaction by • taking the reaction through a different pathway (requiring lower energy) • acting as a surface to “hold” a reactant, to weaken the bonds in the reactant • Why does lowering the activation energy increase the reaction rate?
Catalysis • Energy diagram for catalyzed vs. uncatalyzed reaction:
Catalysis • Energy Distribution Curve: • So the reaction rate increases because with a lower activation energy, the fraction of molecules (collisions) that have the energy needed to react is greater.
Catalysis • Heterogeneous Catalyst • a catalyst that exists in a different phase (usually a solid) from the reactants • often involves gaseous reactants being adsorbed (collected) onto the surface of a solid catalyst • ex: the catalytic hydrogenation of unsaturated hydrocarbons
Catalysis • C2H4(g) + H2(g) ---> C2H6 • normally slow at room temperature • use platinum or palladium as a solid catalyst • H2 and C2H4 are adsorbed on the catalyst surface • the catalyst allows the metal and hydrogen to interact, weakening the H - H single bond
Catalysis • Other examples of heterogeneous catalysts: • SO2 ---> SO3 • ...catalyzed by dust and water droplets • environmentally bad, SO2 is created when sulfur containing fuels are burned, SO3 will react with water to form acid rain • catalytic converters in automobile exhaust engines • converts CO ---> CO2 • hydrocarbons ---> CO2 + H2O • nitric oxide ---> N2 • “poisoned” by lead...hence the need for lead free gasoline
Catalysis • Homogeneous Catalyst • exists in the same phase as the reactants • often reacts with a reactant, but will be reformed in the course of the reaction. • Ex: Catalytic behavior of NO (nitric oxide) and ozone • NO is a pollutant made by automobiles • NO catalyzes ozone production in the troposphere (the layer closest to earth), but catalyzes ozone decomposition in the stratosphere (a layer where ozone is needed for protection against uv).
Catalysis • In the troposphere: • 2NO + O2 --> 2NO2 • 2NO2 --light--> 2NO + 2O • 2O + 2O2 ----> 2O3 (ozone) • Net equation: 3O2 --> 2 O3 • NO does not appear in the overall reaction...it is the catalyst...a substance that reacts, but is reformed • Ozone - an oxidizing agent, toxic, and reacts with other air pollutants to form substances irritating to the eyes and lungs
Catalysis • In the stratosphere: • NO + O3 ---> NO2 + O2 • O + NO2 ---> NO + O2 • Net Equation: O + O3 ---> 2 O2 • So NO again takes part in the reaction, but gets reformed to do its dirty work over and over. • Ozone disappears where its needed, resulting in uv being passing through
Catalysis • More threats to the ozone layer • Freon breaks down in light to make Cl, which catalytically breaks down ozone • (CCl2F2 --light--> CClF2 + Cl) • Cl + O3 ----> ClO + O2 • O + ClO ---> Cl + O2 • Net equation: O + O3 ---> 2O2
Catalysis • Inhibitors • results in a decrease in the reaction rate by • preventing reactants from colliding and reacting • e.g. Paint • reacts preferentially with a reactant • e.g. Anti-oxidants and O2 in potato chips (to prevent the fats from becoming rancid) • knocks out a catalyst • e.g. Lead knocks out the catalyst in a catalytic converter