Kinetics
Kinetics. The Rates and Mechanisms of Chemical Reactions. What does the word KINETIC imply to you? Why should we care about KINETICS? What factors affect KINETICS?. SLOW. FAST. Speed of any event is measured by a change that occurs per unit of time.

Kinetics
E N D
Presentation Transcript
Kinetics The Rates and Mechanisms of Chemical Reactions
What does the word KINETIC imply to you? • Why should we care about KINETICS? • What factors affect KINETICS? SLOW FAST
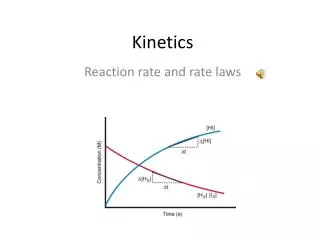
Speed of any event is measured by a change that occurs per unit of time. • The speed of reaction (i.e. reaction rate) is measured as a change in concentration (Molarity; M) of a reactant or product over a certain timescale. • Time is the independent variable (x-axis) and concentration is the dependent variable (y-axis) • Reaction rate is expressed in M/s We Are Talking About Reaction Rates
What happens? • Answer given by balanced chemical equation and stoichiometry • To what extent does it happen? • Answer deals with chemical equilibrium which we will study in a later unit • How fast and by what mechanism? • Chemical kinetics The 3 Fundamental Questions of Chemical Reactions
Chemical Kinetics is very important for biological (your life), environmental (our lives) and economic (industry) processes. • Biological: Large proteins (aka Enzymes) increase the rates of numerous reactions essential to life. • Environmental: The maintenance or depletion of the ozone layer depends on the relative rates of reactions that produce or destroy ozone. • Economic: The synthesis of ammonia (NH3) from N2 and H2 depends on rates of reactions. Fertilizer industries use catalysts to speed up these rates for economic reasons. Why? Importance Examples
Process where energy is released as it proceeds. Heat is given off to surroundings. Reactants Products + Energy Schematic: Exothermic
Process where energy is absorbed as it proceeds. Heat is consumed and surroundings become cooler. Reactants + Energy Products Schematic: Endothermic
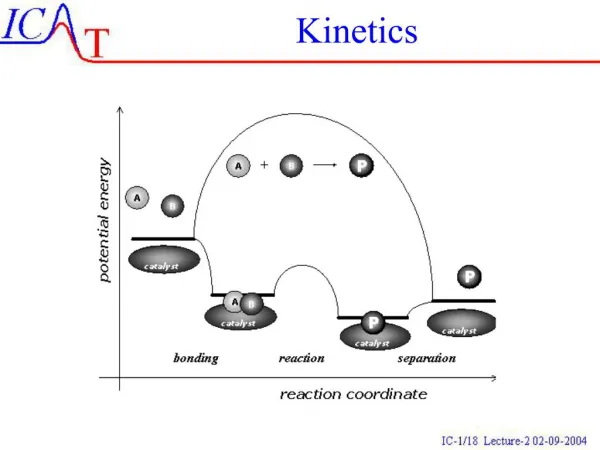
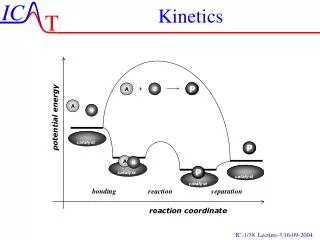
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Concentration of reactants • Temperature • Presence of a catalyst • Surface area • Agitation • Nature of reactants Factors that affect KINETICS
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Concentration of reactants If you increase concentration (Molarity), the rate of reaction increases. Why? There are more molecules which increases the number of collisions altogether; however, there are better chances that molecules will collide in the right orientation. Factor 1: Concentration
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Temperature Temperature is an averaged kinetic energy of molecules so if you increase temperature, you increase kinetic energy. This means you increase the number of collisions Heat supplies the energy to allow the reaction to proceed (i.e. overcoming the activation energy barrier) Think about: Why do we refrigerate milk? Factor 2: Temperature
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Presence of a catalyst Catalyst assist a reaction and increase the reaction rate without being consumed in the reaction. Adding a catalyst decreases the activation energy which means more molecules will have enough energy to react. Think about: Catalytic converter, Enzymes Factor 3: Presence of Catalyst
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Surface Area Increased surface areas of molecules/particles will increase the rate of reaction. This means to break into smaller particle sizes. More places to react give better chances for collisions in the right orientation. How to increase surface area? Grind or crush a mixture of reactants. Ex: A crushed aspirin will enter your blood stream faster than taking it whole. Factor 4: Surface Area
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Agitation Stirring or shaking a reaction will increase the reaction rate. By stirring or shaking, you are introducing energy into the reaction and thus giving molecules/particles more energy to react (overcome activation energy barrier). Your mechanical energy is converted to kinetic energy. Factor 5: Agitation
All based on COLLISION THEORY: Collision theory: For a reaction to occur, the atoms or molecules must collide with one another with enough energy (activation energy) and must collide in the right orientation. FACTORS: • Nature of reactants Reactants whose bonds are weaker have a lower activation energy and thus a higher rate of reaction. All chemical reactions involve bond breaking and bond making. Bond breaking occurs on reactant side. Collisions between reactants that require less kinetic energy are needed to break weaker bonds (i.e. smaller activation energy) Factor 6: Nature of Reactants
a A + b B products ∆[A] ∆t Rate Law: An equation that shows the dependence of the reaction rate on the concentration of each reactant. rate = *[ ] means molarity rate = k[A]m[B]n k is the rate constant. Rate Laws and Reaction Order The values of the exponents (m and n) in the rate law must be determined by experiment; they cannot be deduced from the stoichiometry of the reaction.
∆[NO2] ∆[O2] ∆[N2O5] ∆t ∆t ∆t 2 N2O5(g) aA + bB 4 NO2(g) + O2(g) dD + eE ∆[D] ∆[A] ∆[B] ∆[E] ∆t ∆t ∆t ∆t 1 1 1 1 1 1 b d 4 2 e a – rate = = = General rate of reaction: – – rate = = = = The rate of reaction can be measured based on reactants or products. The negative sign in front for reactants indicates they are consumed. Typically rates of reactions are expressed based on reactants. Rates of Chemical Reactions
ΔT should be Δt (time, not temperature) Rates of Chemical Reactions
The values of the exponents in the rate law must be determined by experiment; they cannot be deduced from the stoichiometry of the reaction. Rate Laws and Reaction Order
2 NO(g) + O2(g) 2 NO2(g) rate = k[NO]m [O2]n Compare the initial rates to the changes in initial concentrations. Determining a Rate Law: The Method of Initial Rates
2 NO(g) + O2(g) 2 NO2(g) rate = k[NO]2 [O2]n The concentration of NO doubles, the concentration of O2 remains constant, and the rate quadruples. 2m = 4 m = 2 Determining a Rate Law: The Method of Initial Rates
2 NO(g) + O2(g) 2 NO2(g) rate = k[NO]2 [O2] Reaction Order with Respect to a Reactant • NO: second order • O2: first order Overall Reaction Order • 2 + 1 = 3 (third order) Determining a Rate Law: The Method of Initial Rates
2 NO(g) + O2(g) 2 NO2(g) 1 M2 s rate M rate = k[NO]2 [O2] s [NO]2 [O2] Units for this third-order reaction: k = = = (M2) (M) Determining a Rate Law: The Method of Initial Rates You can pick any experiment or trial and solve for k (should all be the same).
[HgCl2] [C2O42-] Rate (M/s) 0.164 0.15 3.2 x 10-5 0.164 0.45 2.9 x 10-4 0.082 0.45 1.4 x 10-4 • Determine the rate law. What is the order of the reaction? • Determine the rate law constant (specify the units) • What is the rate when the initial concentrations of both reactants are 0.100 M? Example Problem #1
[HgCl2] [C2O42-] Rate (M/s) 0.164 0.15 3.2 x 10-5 0.164 0.45 2.9 x 10-4 0.082 0.45 1.4 x 10-4 • Determine the rate law. What is the order of the reaction? Rate = k[HgCl2]m[C2O42-]n = = = , = = = , Rate = k[HgCl2][C2O42-]2 Overall third order Example Problem #1
[HgCl2] [C2O42-] Rate (M/s) 0.164 0.15 3.2 x 10-5 0.164 0.45 2.9 x 10-4 0.082 0.45 1.4 x 10-4 • Determine the rate law. What is the order of the reaction? • Determine the rate law constant (specify the units) Rate = k[HgCl2][C2O42-]2 3.2x10-5M/s = k[0.164M][0.15M]2 = = Example Problem #1
[HgCl2] [C2O42-] Rate (M/s) 0.164 0.15 3.2 x 10-5 0.164 0.45 2.9 x 10-4 0.082 0.45 1.4 x 10-4 • Determine the rate law. What is the order of the reaction? • Determine the rate law constant (specify the units) • What is the rate when the initial concentrations of both reactants are 0.100 M? Rate = k[HgCl2][C2O42-]2 )(0.100M)(0.100M)2 = Example Problem #1
[NOCl] Rate M/s 3000 5980 2000 2660 1000 665 4000 10640 • Determine the rate law and order. • Determine the rate law constant. Specify the units. • What is the rate when the concentration of NOCl is 8000M? Example Problem #2
[NOCl] Rate M/s 3000 5980 2000 2660 1000 665 4000 10640 Determine the rate law and order. Rate = k[NOCl]2; Second Order Determine the rate law constant. Specify the units. For first trial: 6.64x10-4(1/Ms) For second trial: 6.65x10-4(1/Ms) What is the rate when the concentration of NOCl is 8000M? 42560 M/s Example Problem #2
[A] [B] Rate (M/s) 1.50 1.50 0.32 1.50 2.50 0.32 3.00 1.50 0.64 • Determine the rate law and order. • Determine the rate law constant. Specify the units. Example #3
[A] [B] Rate (M/s) 1.50 1.50 0.32 1.50 2.50 0.32 3.00 1.50 0.64 Determine the rate law and order. Rate = k[A]; first order Determine the rate law constant. Specify the units. k = 0.21 (1/s) Example #3
[CH3COCH3] [Br2] [H+] Rate (M/s) 0.30 0.05 0.05 0.000057 0.30 0.10 0.05 0.000057 0.30 0.05 0.10 0.000120 0.40 0.05 0.20 0.000310 0.40 0.05 0.05 0.000076 • Determine the rate law and order. • Determine the rate law constant. Specify the units. Example #4
[CH3COCH3] [Br2] [H+] Rate (M/s) 0.30 0.05 0.05 0.000057 0.30 0.10 0.05 0.000057 0.30 0.05 0.10 0.000120 0.40 0.05 0.20 0.000310 0.40 0.05 0.05 0.000076 Determine the rate law and order. Rate = k[CH3COCH3][H+]; second order Determine the rate law constant. Specify the units. k = 0.0038 (1/Ms) Example #4
[S2O8-2] [I -] Rate (mol/L/s) 0.018 0.036 2.6 x 10-6 0.027 0.036 3.9 x 10-6 0.036 0.054 7.8 x 10-6 0.050 0.072 1.4 x 10-5 • Determine the rate law and order. • Determine the rate law constant. Specify the units. Example #5
[S2O8-2] [I -] Rate (mol/L/s) 0.018 0.036 2.6 x 10-6 0.027 0.036 3.9 x 10-6 0.036 0.054 7.8 x 10-6 0.050 0.072 1.4 x 10-5 Determine the rate law and order. Rate = k[S2O8-2]m[I-]n = = = , = = = , Rate = k[S2O8-2][I-] Second order Determine the rate law constant. Specify the units. For first trial: 0.0040 (1/Ms) For second trial: 0.0040 (1/Ms) Example #5
Integrated Rate Laws Graphs, reaction order and concentrations.
Scientists don’t like curves, they like straight lines. SO they have to manipulate to obtain straight line. Graphs: concentration of substance A vs. time
Determining a Rate Law: The Method of Initial Rates *Units for reaction rates are always M/s
A product(s) ∆[A] – = k ∆t For a zeroth-order reaction, the rate is independent of the concentration of the reactant. t½= [A]0 2k rate = k[A]0 = k Calculus can be used to derive an integrated rate law. [A]t concentration of A at time t [A]0 initial concentration of A [A]t = –kt + [A]0 Zeroth-Order Reactions y = mx + b
A plot of [A] versus time gives a straight-line fit and the slope will be –k. Zeroth-Order Reactions
For zeroth order: [A]t = –kt + [A]0 [A]t1/2= [A]0 2 • Half-life: time for A to reduce by 50% = - = Half-Life: t1/2
A product(s) [A]t x = –kt ln ln = ln(x) – ln(y) [A]0 y ∆[A] – = k[A] ∆t rate = k[A] Calculus can be used to derive an integrated rate law. [A]t concentration of A at time t [A]0 initial concentration of A First-Order Reactions: The Integrated Rate Law ln[A]t = –kt + ln[A]0 Using: y = mx + b
ln[A]t = –kt + ln[A]0 First-Order Reactions: The Integrated Rate Law
A product(s) 1 = –kt1/2 ln [A]t 2 = –kt ln [A]0 [A]0 = [A] 0.693 t1/2 2 t1/2 = k Half-Life: The time required for the reactant concentration to drop to one-half of its initial value. rate = k[A] t = t1/2 First-Order Reactions: Half-Life or
A product(s) = kt + 1 1 [A]t [A]0 ∆[A] – = k[A]2 ∆t rate = k[A]2 Calculus can be used to derive an integrated rate law. [A]t concentration of A at time t [A]0 initial concentration of A y = mx + b Second-Order Reactions
2 NO2(g) 2 NO(g) + O2(g) Second-Order Reactions
2 NO2(g) 2 NO(g) + O2(g) Second-Order Reactions
A product(s) = kt1/2 + = kt + 1 1 2 1 t1/2 = [A]0 [A]0 [A]0 [A]t [A]0 1 = [A] t1/2 k[A]0 2 Half-life for a second-order reaction: rate = k[A]2 t = t1/2 Second-Order Reactions
t1/2 = 1 k[A]0 For a second-order reaction, the half-life is dependent on the initial concentration. Each successive half-life is twice as long as the preceding one. Second-Order Reactions