Kinetics
Kinetics. The study of reaction rates. Spontaneous reactions are reactions that will happen - but we can’t tell how fast. Diamond will spontaneously turn to graphite – eventually. Reaction mechanism- a series of steps by which a reaction takes place. Reaction Rate.

Kinetics
E N D
Presentation Transcript
Kinetics • The study of reaction rates. • Spontaneous reactions are reactions that will happen - but we can’t tell how fast. • Diamond will spontaneously turn to graphite – eventually. • Reaction mechanism- a series of steps by which a reaction takes place.
Reaction Rate • Rate = [A] at time t2 - [A] at time t1t2 – t1 • Rate =D[A]Dt • Reaction Rate – is a change in concentration of a reactant or product per unit time • For this reaction: • 2NO2 (g) → 2NO (g) + O2 (g)
Reaction Rates: 2NO2(g) 2NO(g) + O2(g) 1. Can measure disappearance of reactants 2. Can measure appearance of products 3. Are proportional stoichiometrically
Reaction Rates: 2NO2(g) 2NO(g) + O2(g) 4. Are equal to the slope tangent to that point 5. Change as the reaction proceeds, if the rate is dependent upon concentration [NO2] t
Defining Rate of a Reaction • We can define rate in terms of the disappearance of the reactant or in terms of the rate of appearance of the product. • In our example N2 + 3H2 2NH3 • -D[N2] or -3D[H2] or 2D[NH3] Dt Dt Dt
Rate Laws • Reactions are reversible. • As products accumulate they can begin to turn back into reactants. • Early on the rate will depend on only the amount of reactants present. • We want to measure the reactants as soon as they are mixed. • This is called the Initial rate method.
Rate Laws • Two key points • The concentration of the products do not appear in the rate law because this is an initial rate. • The order must be determined experimentally, it cannot be obtained from the equation.
2 NO2(g) →2NO(g) + O2(g) • Rate will only depend on the concentration of the reactants. Products have not built up yet. To define rate in terms of NO2: • Rate = k[NO2]n • This is called a rate law expression. • k is called the rate constant. • n is the order of the reactant - usually a positive integer. (must be determined by experiment)
Types of Rate Laws • Differential Rate law - describes how rate depends on concentration. (Typically just called Rate Law) • Integrated Rate Law - Describes how concentration depends on time. • For each type of differential rate law there is an integrated rate law and vice versa. • Rate laws can help us better understand reaction mechanisms.
Determining Rate Laws • The first step is to determine the form of the rate law (especially its order). • Must be determined from experimental data. • For this reaction in CCl4 soln. 2 N2O5(aq) → 4NO2(aq) + O2 (g)Oxygen escapes the solution and therefore there is no reverse reaction
[N2O5] (mol/L) Time (s) 1.00 0 0.88 200 0.78 400 0.69 600 0.61 800 0.54 1000 0.48 1200 0.43 1400 0.38 1600 0.34 1800 0.30 2000 Now graph the data
To find rate we have to find the slope at two points • We will use the tangent method.
.90 = -5.4 x 10-4 .45 -2.7 x 10-4 • As the [ ] doubles the rate of reaction doubles. (Twice as fast) • Rate = -D[N2O5] = k[N2O5]1 = k[N2O5] Dt • We say this reaction is first order in N2O5
The method of Initial Rates • This method requires that a reaction be run several times. • The initial concentrations of the reactants are varied. • The reaction rate is measured just after the reactants are mixed. • Eliminates the effect of the reverse reaction.
An example • For the reaction BrO3- + 5 Br- + 6H+ 3Br2 + 3 H2O • The general form of the Rate Law is Rate = k[BrO3-]n[Br-]m[H+]p • We use experimental data to determine the values of n,m,and p
Initial concentrations (M) Rate (M/s) Now we have to see how the rate changes with concentration BrO3- Br- H+ 0.10 0.10 0.10 8.0 x 10-4 0.20 0.10 0.10 1.6 x 10-3 0.20 0.20 0.10 3.2 x 10-3 0.10 0.10 0.20 3.2 x 10-3
Integrated Rate Law • Expresses the reaction concentration as a function of time. • Form of the equation depends on the order of the rate law (differential). • Changes Rate = D[A]nDt • We will only work with n=0, 1, and 2
1st Order Rate Laws • For the reaction 2N2O5 4NO2 + O2 • We found the Rate = k[N2O5]1 • If concentration doubles rate doubles. • If we integrate this equation with respect to time we get the Integrated Rate Law • ln[N2O5] = - kt + ln[N2O5]0 • ln is the natural log • [N2O5]0 is the initial concentration.
1st Order – Important Points • Equation shows how conc. of A depends on time. • General form Rate = D[A] / Dt = k[A] • ln[A] = - kt + ln[A]0 • In the form y = mx + b • y = ln[A] m = -k • x = t b = ln[A]0 • A graph of ln[A] vs time is a straight line.
First Order • By getting the straight line you can prove it is first order • Often expressed in a ratio
First Order • By getting the straight line you can prove it is first order • Often expressed in a ratio
Example 12.2 • The decomposition of N2O5 in the gas phase was studied at constant temperature • 2N2O5 (g) → 4NO2 (g) + O2 (g) • [N2O5] (mol/L) Time (s) 0.1000 0 0.0707 50 0.0500 100 0.0250 200 0.0125 300 0.00625 400 Verify the rate law is 1st order and calculate the rate constant
Example 12.3 • Using the data given in Example 12.2, calculate [N2O5] at 150s after the start of the reaction.
Half Life • The time required to reach half the original concentration. • If the reaction is first order • [A] = [A]0/2 when t = t1/2
Half Life • The time required to reach half the original concentration. • If the reaction is first order • [A] = [A]0/2 when t = t1/2 • ln(2) = kt1/2
Half Life • t1/2 = 0.693/k • The time to reach half the original concentration does not depend on the starting concentration. • An easy way to find k
Example 12.4 • A certain first-order reaction has a half-life of 20.0 minutes. • a. calculate the rate constant for this reaction • b. how much time is required for this reaction to be 75% complete?
Second Order • Rate = -D[A] / Dt = k[A]2 • integrated rate law • 1/[A] = kt + 1/[A]0 • y= 1/[A] m = k • x= t b = 1/[A]0 • A straight line if 1/[A] vs t is graphed • Knowing k and [A]0 you can calculate [A] at any time t
Second Order Half Life • [A] = [A]0 /2 at t = t1/2
Example 12.5 • Butadiene reacts to form its dimer according to the equation • 2C4H6 (g) → C8H12 (g) [C4H6] (mol/L)Time (s) 0.01000 0 0.00625 1000 0.00476 1800 0.00370 2800 0.00313 3600 0.00270 4400 0.00241 5200 0.00208 6200 • Is this reaction first or second order? • What is the value of the rate constant for the reaction? • What is the half life for the reaction
Integrated Rate Law • For the reaction: • NO2 (g) + CO (g) → NO (g) + CO2 (g) • Time (s)[NO2] (mol/L) • 0 0.500 • 1.20 x 103 0.444 • 3.00 x 103 0.381 • 4.50 x 103 0.340 • 9.00 x 103 0.250 • 1.80 x 104 0.174 • 1. Determine the rate law and the integrated rate law • 2. Calculate the rate constant • 3. Calculate [NO2] at 2.70 x 104 s from start of reaction.
Zero Order Rate Law • Rate = k[A]0 = k • Rate does not change with concentration. • Integrated [A] = -kt + [A]0 • When [A] = [A]0 /2 t = t1/2 • t1/2 = [A]0 /2k
Zero Order Rate Law • Most often when reaction happens on a surface because the surface area stays constant. • Also applies to enzyme chemistry.
Summary of Rate Laws • We study under conditions where only the forward reaction is important • Two types of rate laws • Differential rate law – how rate depends on concentration • Integrated rate law – how concentration depends on time • Which rate law is used depends on the type of data that can be collected conveniently • For full summary read Sect. 12.5 on p. 548
Reaction Mechanisms • The reaction mechanism is the series of elementary steps by which a chemical reaction occurs\ • Must satisfy the following two requirements • (1) The sum of the elementary steps must give the overall balanced equation for the reaction • (2) The mechanism must agree with the experimentally determined rate law
Rate Determining Step • In a multi-step reaction, the slowest step is the rate-determining step (RDS). It therefore determines the rate of the reaction. • The experimental rate law must agree with the rate-determining step
Identifying the Rate Determining Step • For the reaction: • 2H2(g) + 2NO(g) N2(g) + 2H2O(g) • The experimental rate law is: • R = k[NO]2[H2] • Which step in the reaction mechanism is the rate-determining (slowest) step? • Step #1 H2(g) + 2NO(g) N2O(g) + H2O(g) • Step #2 N2O(g) + H2(g) N2(g) + H2O(g) • Step #1 agrees with the experimental rate law
Validity of a Mechanism • 2NO2 + F2 2NO2F • Rate = k[NO2][F2] • The proposed mechanism is • NO2 + F2 NO2F + F (slow) • F + NO2 NO2F (fast) • F is called an intermediate It is formed then consumed in the reaction • Mechanism is valid if: Rate law from RDS matches the overall rate law.
Reaction Mechanisms • Each of the two reactions is called an elementary step. • The rate for a reaction can be written from its molecularity . • Molecularity is the number of pieces that must come together.
Rate Laws from elementary steps • A products Rate = k[A] • A+A products Rate= k[A]2 • 2A products Rate= k[A]2 • A+B products Rate= k[A][B] • A+A+B Products Rate= k[A]2[B] • 2A+B Products Rate= k[A]2[B] • A+B+C Products Rate= k[A][B][C]
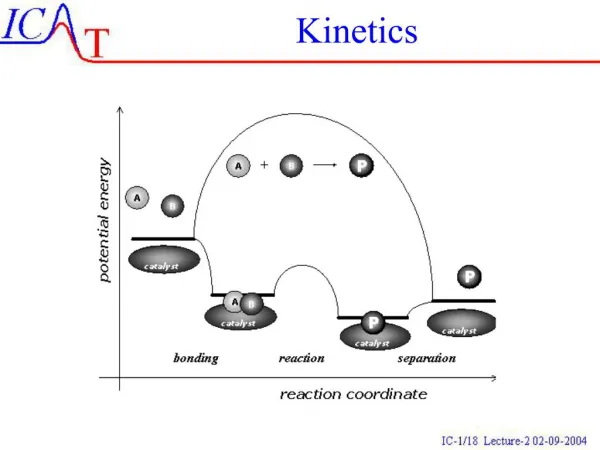
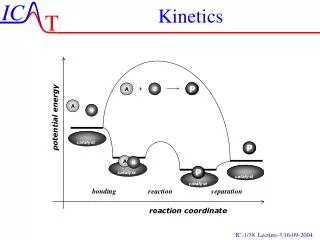
Collision theory • Molecules must collide to react. • Concentration affects rates because collisions are more likely. • Collisions must have sufficient energy to produce the reaction (must be = or > the activation energy). • Colliding particles must be correctly oriented to one another in order to produce a reaction. • Only a small number of collisions produce reactions.
Factors Affecting Rate • Increasing temperaturealways increases the rate of a reaction. • Particles collide more frequently • Particles collide more energetically • Increasing surface area increases the rate of a reaction • Increasing ConcentrationUSUALLY increases the rate of a reaction • Presence of Catalysts, which lower the activation energy by providing alternate pathways
Potential Energy Reactants Products Reaction Coordinate
Potential Energy Activation Energy Ea Reactants Products Reaction Coordinate
Activated complex Potential Energy Reactants Products Reaction Coordinate
Potential Energy } Reactants DE Products Reaction Coordinate
Br---NO Potential Energy Br---NO Transition State 2BrNO 2NO + Br 2 Reaction Coordinate
Terms • Activation energy - the minimum energy needed to make a reaction happen. • Activated Complex or Transition State - The arrangement of atoms at the top of the energy barrier.