Organic Molecules
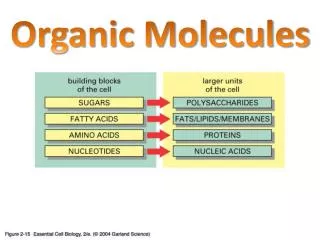
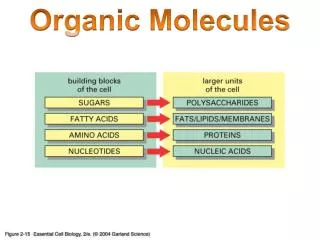
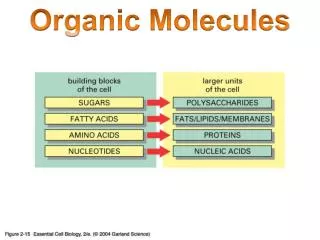
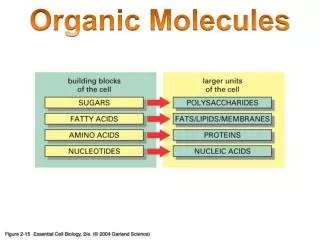
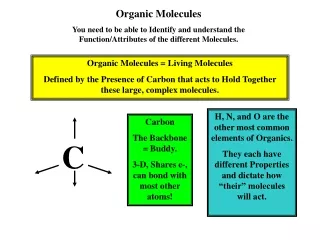
Organic Molecules. Organic ≡ Carbon based Most numerous class of materials Countless combinations are possible Carbon bonds with almost any other element Carbon to carbon bonds very common Gases (acetylene, propane) Linear chain liquids (gasoline, oil)

Organic Molecules
E N D
Presentation Transcript
Organic Molecules • Organic ≡ Carbon based • Most numerous class of materials • Countless combinations are possible • Carbon bonds with almost any other element • Carbon to carbon bonds very common • Gases (acetylene, propane) • Linear chain liquids (gasoline, oil) • Ring structures (benzene, cyclopentane) • Crystalline structures (diamond, graphite)
Hydrocarbons • Alkane series, linear progression of carbon • Methane = 1 carbon (natural gas) • Ethane = 2 carbon (ripening fruit) • Propane = 3 carbon chain (gas BBQ) • Butane = 4 carbon chain (cigarette lighters) • Pentane = 5 carbon chain • Hexane = 6 carbon chain (Naptha) • Heptane = 7 carbon chain • Octane = 8 carbon chain (gasoline reference)
Naptha & KeroseneNaptha is less dense than gasolineKerosene is heavier (same as Jet Fuel)
Chemically very similar Home heating oil can be used in diesel cars, and vice versa Fuel taxed by application Road Tax for highway use, not home heating Northeastern states put dye color in heating oil Don’t get caught with colored fuel in your car! Diesel = Heating Oil Legal Issue
Longer molecule than gasoline, more carbon bonds, greater mileage Diesel engines simpler, more robust & reliable Older fuels had more sulfur, SO2 into the air Older design engines also made lots of soot. Fuels now cleaner, and cars are more efficient. Europe about 50/50 gasoline vs diesel cars Diesel making a comeback
Fractional Distillation of Crude Oilmultitude of carbon lengths & shapes from natural processesDiesel fuel contains >150 different molecules
Hydrocarbons • Linear Hydrocarbons • “backbone” not branched, continuous (zig-zag) line • Branched Hydrocarbons • Molecule has branches, usual term is “iso” • Iso-octane, Isopropyl • Ring structures • Carbon backbone closed in on itself • “aliphatic” no double bonds (e.g. cyclohexane) • “aromatic” with double bonds (e.g. benzene) • “Buckyballs” are closed hollow surfaces (e.g. C60)
Octane and Iso-OctaneZig-Zag shape dictated by tetrahedral bonding
Oxygen based attachments • Oxygen between carbons is an “ether” • Ethyl ether is H3C-O-CH3 • Oxygen to side of carbon is a “ketone” • Acetone is H3C-CO-CH3 • Hydroxyl on carbon is “alcohol” • Ethanol is C2H5OH • Carbon with 2 oxygen is “Carboxyl” • Carboxylic Acid (e.g. vinegar = Acetic Acid) • Alcohol + Carboxylic acid = “ester” • Basis of many flavors and odors
EthersEthers identified by central “–O-”group • Diethyl Ether (or Ethyl Ether) • an early general anesthetic, first used 1842 • Low boiling point, extremely flammable • Used as automotive “starting fluid” p. 368
KetonesKetones contain branched double bond oxygen on carbon • Acetone is simplest ketone • A strong solvent, nail polish remover • Mixes with water or oil
AlcoholsAlcohols identified by “-OH” group • Ethanol, component of alcoholic beverages • Now considered an “alternative” to Gasoline • E-85 is 85% ethanol, 15% gasoline • Some stations in San Jose now offer it p. 368
E15 motor fuel • E15 contains 15% ethanol and 85% gasoline. This is generally the greatest ratio of ethanol to gas that is recommended by auto manufacturers that sell vehicles in the United States, though it is possible that many vehicles can handle higher mixtures without trouble. Flexible-fuel vehicles are designed to take higher concentrations, up to 96% v/v ethanol (and no gasoline).
E20 motor fuel • E20 contains 20% ethanol and 80% gasoline. This fuel is not yet widely used in the United States, but will be mandated by the U.S. state of Minnesota by 2013. Since February 2006, this is the standard ethanol-gasoline mixture sold in Brazil, where concerns with the alcohol supply resulted in a drop in the ethanol percentage, previously at 25%. Flexible-fuel cars are set up to run with gasoline in such concentration range and few will work properly with lower concentrations of ethanol
E85 motor fuel • E85 is a mixture of 85% ethanol and 15% gasoline, and is generally the highest ethanol fuel mixture found in the United States. It is common in Sweden, and there are more than 1000 public E85 fuel pumps in the U.S. as of 2006, mostly concentrated in the Midwest, with over half of those in Minnesota. • This mixture has an octane rating of about 105. This is down significantly from pure ethanol but still much higher than normal gasoline. The addition of a small amount of gasoline helps a conventional engine start when using this fuel under cold conditions.
“Flex-Fuel” cars Operate with multiple fuels Alcohol or Gasoline in Brazil Both available at same station Motorists buy cheapest one Alcohol has 70% mileage of gas Alcohol 70% price = breakeven Bring your calculator ! Local availability of E-85
CarboxylatesIdentified by “-COOH” group (acid form) • Formic Acid, H-COOH, bee-sting venom • Acetic acid (vinegar) is H3C-COOH • Propionic Acid (preservative) H3C-CH2-COOH • Butyric Acid (rancid butter) H3C-CH2-CH2-COOH
Carboxylic Acid ionizationHydrogen ion + Carboxylate ion • Acetic acid ionizes to hydrogen + acetate ion P/review, p. 385
Computer Modeling • CABRILLO COLLEGE has an interactive molecule display, allowing users to twist and turn common molecules to investigate their structures. Almost like a video game, but more educational than killing aliens. We sometimes use this in lab classes, lots of options to play around with. • http://c4.cabrillo.edu/chem30a/exercises/Exer_1/index.html • http://c4.cabrillo.edu/chem30b/exercises/chpt_11_060700/index.html