Dilution
Dilution. Dilution. Adding water to a particular stock solution to achieve a desired molarity. Dilution. Moles of solute after dilution = Moles of solute before dilution Only water is added! Solute is not added or subtracted !. Dilution. MV = MV

Dilution
E N D
Presentation Transcript
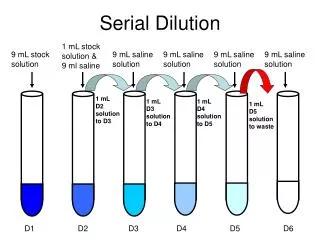
Dilution • Adding water to a particular stock solution to achieve a desired molarity
Dilution • Moles of solute after dilution = Moles of solute before dilution • Only water is added! Solute is not added or subtracted !
Dilution • MV = MV • What volume of 16M H2SO4 must be used to prepare 1.5 L of 0.10 M H2SO4 solution?
Types of Chemical Rxns • Precipitation rxns • Acid/Base rxns • Oxidation/Reduction rxns
Precipitation Rxns • When two solutions are mixed and an insoluble substance forms • The solid is the precipitate
Precipitation Rxns • Best way to predict products is to think about what products are possible
Precipitation Rxns • In almost every case, when a solid containing ions dissolves in H2O, the ions separate and move independently.
Hints that help... • 1. When ions form a solid, the compound must have a 0 net charge. (i.e. the products must contain a cation and anion)
Hints that help... • 2. Most ionic materials contain only one type of cation and one type of anion.
A rxn to try... • K2CrO4(aq) + Ba(NO3)2(aq) -->products?? • The product includes a yellow solid...
Possible ions... • K+ CrO42- Ba2+ NO3- • Possible products • K2CrO4 KNO3 BaCrO4 Ba(NO3)2
Only real possibilities... • KNO3 and BaCrO4 • An experienced chemist has more data-- CrO4 is a yellow ion
The final result... • K2CrO4(aq) + Ba(NO3)2(aq) --> BaCrO4(s) + 2KNO3(aq)
Solubility Rules for Salt • 1. Nitrate (NO3-) salts are soluble.
Solubility Rules for Salt • 2. Salts containing alkali metal ions (Li+, Na+, K+, Cs+, Rb+) and the ammonium ion (NH4+) are soluble.
Solubility Rules for Salt • 3. Most chloride, bromide, and iodide salts are soluble. Notable exceptions are salts with ions Ag+, Pb2+, and Hg22+
Solubility Rules for Salt • 4. Most sulfate salts are soluble. Notable exceptions are BaSO4, PbSO4, HgSO4, and CaSO4.
Solubility Rules for Salt • Most hydroxide salts precipitate (or dissolve only slightly). Impt soluble hydroxides are NaOH and KOH.
Solubility Rules for Salt • Ba(OH)2, Sr(OH)2 and Ca(OH)2 are marginally soluble.
Solubility Rules for Salt • Most sulfide (S2-), carbonate (CO32-) and phosphate (PO43-) salts precipitate.
Types of Equations • Molecular equation • shows the reactants and products of a reaction
Molecular Eqn Example • AgNO3(aq) + KCl(aq) --> AgCl(s) + KNO3(aq) • Does not give clear picture of what occurs!
Complete Ionic Eqn • All substances that are strong electrolytes (ionized) are represented as ions
Complete Ionic Eqn • Ag+(aq) + NO3-(aq) + K+(aq) + Cl-(aq)-->AgCl(s) + K+(aq) + NO3-(aq)
Notice! • The K+ and the NO3- ions do not participate in the reaction!!! • They are spectator ions.
Net Ionic Eqns • Eliminate the spectator ions from the complete ionic equation.
Net Ionic Eqns • Ag+(aq) + Cl-(aq) --> AgCl(s)
Stoichiometry of ppt rxn • Same as stoichiometry that we already learned! • Must figure out rxn first! • Must get moles from M!