Chapter 18 Lipids
Chapter 18 Lipids. 18.1 Lipids 18.2 Fatty Acids. Lipids. Lipids are: Biomolecules that contain fatty acids or a steroid nucleus. Soluble in organic solvents, but not in water. Named for the Greek word lipos , which means “fat.” Extracted from cells using organic solvents.

Chapter 18 Lipids
E N D
Presentation Transcript
Chapter 18 Lipids 18.1 Lipids 18.2 Fatty Acids
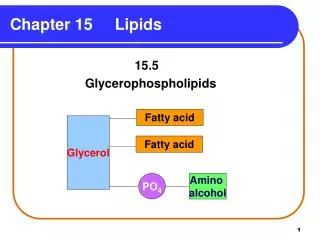
Lipids Lipids are: • Biomolecules that contain fatty acids or a steroid nucleus. • Soluble in organic solvents, but not in water. • Named for the Greek word lipos, which means “fat.” • Extracted from cells using organic solvents.
Diversity in structure leads to a diversity in biological function • Non-polar lipids (fats) are the principal molecules for energy storage • Polar lipids (nitrogen and phosphorus containing) are components of biological membranes • The steroid class of lipids (cholesterol) is found in biological membranes and used as a precursor for many hormones • Miscellaneous lipids present only in minor quantities in the cell are involved as light-absorbing pigments (β-carotene, retinal), enzyme cofactors (vitamin K), signal molecules (prostaglandins), and electron carries (ubiquinone)
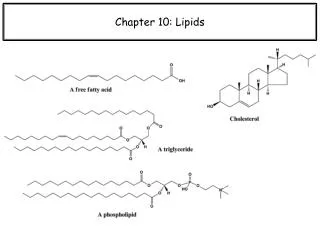
Fatty Acids Fatty acids are: • Long-chain carboxylic acids that have an even number (usually 12-18) of carbon atoms. • Insoluble in water. • Saturated or unsaturated.
Fatty Acid Formulas • The formulas for fatty acids are written as condensed and line-bond formulas. For example caprylic acid with 8 carbon atoms can be written as: CH3—(CH2)6—COOH CH3—CH2—CH2—CH2—CH2—CH2—CH2—COOH O || CH3—CH2—CH2—CH2—CH2—CH2—CH2—C—OH
Ionization • General fatty acid formula: • R – COOH (R stands for alkyl chain composed of carbon and hydrogen atoms) • The pKa of the fatty acid carboxyl group is about 4.8 • Under ordinary conditions the pH of the plasma is 7.4, and the pH of the intracellular fluid is about 7.0 • Almost all (99.9%) of the free fatty acid molecules in body fluids are ionized (the fatty acid is present as an anion) • RCOOH ↔ RCOO- + H+ • The ionized group interacts with aqueous media (polar) • The hydrocarbon “tail” seeks a non-polar environment
General nomenclature • The carbon atoms of an acid are numbered (or lettered with Greek lettering system, such as α, β, γ) either from the carboxyl group or from the carbon farthest removed from the carboxyl group (n or omega numbering system) • CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH2-COOH • 10 9 8 7 6 5 4 3 2 1 • 1 2 3 4 5 6 7 8 9 10 • ωδγβα
Saturated Fatty Acids Saturated fatty acids have single C–C bonds.
Saturated Fatty Acids Saturated fatty acids have: • Molecules that fit closely together in a regular pattern. • Strong attractions between fatty acid chains. • High melting points that makes them solids at room temperature.
Unsaturated Fatty Acids • Unsaturated fatty acids typically contain cis double bonds.
Unsaturated Fatty Acids Unsaturated fatty acids have cis C=C bonds.
Unsaturated Fatty Acids Unsaturated fatty acids: • Have nonlinear chains that do not allow molecules to pack closely. • Have low melting points. • Are liquids at room temperature.
Olive Oil • Olive oil contains a high percentage of oleic acid. • Oleic acid is a monounsaturated fatty acid with one cis double bond.
Melting Points of Fats and Oils Vegetable oils contain more unsaturated fatty acids, which makes their melting points lower.
Canola oil • Canola Oil - Truth and Myths about Canola • http://www.canola-council.org/about/thetruth.html
Omega-6 and Omega-3 Fatty Acids The first double bond: • In vegetable oils is at carbon 6 (omega-6). • In fish oils is at carbon 3 (omega-3).
Essential fatty acids • Mammals cannot synthesize the two main types of polyunsaturated fatty acids: the linoleic (omega-6) and linolenic (omega-3) • Essential fatty acid deficiency is characterized by dermatitis and poor wound healing • Most prostaglandins are synthesized from arachidonic acid (a linoleic acid derivative) • Lack of these prostaglandins causes some aspects of the essential fatty acid deficiency syndrome • A small daily intake of linolenic acid is recommended (this may be especially important when the nervous system is developing) • Docosahexaenoic acid (22:6n-3) – omega-3 fatty acid is needed for maximum development of the visual and central nervous systems during the neonatal period (it is present in large amounts in the retina and parts of the brain; it enhances the visual response and the function of certain domains in neural membranes)
Prostaglandins Prostaglandins have: • 20 carbon atoms in their fatty acid chains. • An OH on carbon 11 and 15. • A trans double bond at carbon 13.
Prostaglandins in the Body Prostaglandins are: • Produced by injured tissues. • Involved in pain, fever, and inflammation. • Not produced when anti-inflammatory drugs such as aspirin inhibit their synthesis. • Prostaglandins • http://www.elmhurst.edu/~chm/vchembook/555prostagland.html
Other eicosanoids • When a cell responds to a stimulus, bioactive mediators (prostaglandins, thromboxanes, leukotrienes, and others) often are formed from the lipids contained in its membranes. • Thromboxanes regulate blood coagulation. • Leukotrienes play a role in allergic and inflammation responses. They were isolated from leukocytes (white blood cells).