Hepatitis B Virus
Hepatitis B Virus. Historical Background. Only human and chimpanzee-parenteraly S protein of HBV 1963 B.S. Blumberg D.S. Dane – 42nm endogenous DNA polymerase activity 4. 1979 – cloning and sequencing S, C, P, X 5. Chronic hepatitis, hepatocellular carcinoma

Hepatitis B Virus
E N D
Presentation Transcript
Historical Background • Only human and chimpanzee-parenteraly • S protein of HBV 1963 B.S. Blumberg • D.S. Dane – 42nm endogenous DNA polymerase activity 4. 1979 – cloning and sequencing S, C, P, X 5. Chronic hepatitis, hepatocellular carcinoma 6. Noncytocidal infection of hepatocyte
Mode of Transmission & Course of Disease of Different Hepadnaviruses
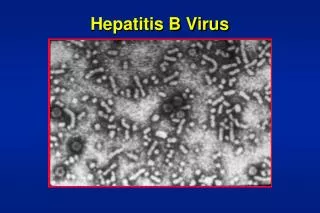
Morphology of Purified HBV and HBs Particles A B C A. HBV is spherical with double shelled structure. B. When largest of the three HBs proteins is overproduced, filamentous particles are formed. C. HBs particles appear mostly as sphere particles.
Hepatotropism • level of viral gene expression • Reservoir for virus to persist • Phagocytic activity detection of viral nucleic acid • Mediated by pre S1 • HBsAg filaments (rich in LHBs), 20nm particle (mainly SHBs) • Inhibition of binding with Mab (27-49 AAs) • Pre S2 supportive function Cellular receptor membrane -bound interleukin-6 SHBs -Apolipoprotein H, endonexin II no signal receptor
Viral Genome • Relaxed circular, partially duplex DNA • Between 3,182 to 3,221 bases long • P protein : covalently binding to 5' end of minus strand • 2 direct repeat (DR1, DR2) • Compact genome with ORFs encoding multiple proteins • 4 ORF ( C, P, PreS/S, X ) • Contains numerous genetic elements that regulate transcription
Viral Replication 1. Genome is transported to the nucleus and converted cccDNA. 2. Three essential classes of mRNA are transcribed. 3. Translation of core/pol transcript allows for assembly of core particles containing pregenome. 4. LHB and M/SHBs transcripts are translated at rough ER and HBs proteins are inserted into membrane. 5. HBs particles bud to lumen and envelope core particles. 6. HBV and HBs particles are secreted. HBV life cycle
Viral uptake Long-term cultivated hepatocytes • not susceptible but productive • Primary hepatocyte culture • Increase copy number of intercellular viral DNA • Endocytotic pathway (DHBV uptake) no data for HBV • Susceptibility with protease (HepG2) pre S2 domain
Nuclear Entry of the Viral Genomes • Core particle in the nuclear (30nm 34nm diameter) – upper limit of nuclearpore 30nm • Nuclear localization signal of the carboxyl terminal of the core molecule ( fig. ) • Transfected cell – 1/1000 nuclear DNA • Viral polymerase – mediated transport of the viral genome • Amino- or/and carboxyl truncated pol – much more efficient
Schematic Diagram of Intracellular Life Cycle of HBV Core Particle and HBV Genome • 1 & 2. Assembly of RNA, pol, PKC and • Core subunits. • 3. Pol Synthesizes minus DNA strand • and PKC phosphorylates core • subunits. • Structural change occurs in core subunit, NBS is exposed • NBS is bound by importins • 6. & 7. Core binds to NPC and • disintegrates • 8. Pol and DNA complex are imported to nucleus and DNA is converted to cccDNA
Formation of Covalently Closed Circular (ccc) DNA • cccDNA is the template for the transcription of viral mRNA • cccDNA detected with in the first 24h • Prior to cccDNA • Removal of the covalently bound polymerase • Removal of RNA primer • Completion of (+)DNA strand • Repair and ligation of the gaps – maybe cellular DNA polymerase is essential Core could also act as a vector for the nuclear transport of viral DNA cccDNA accumulates only in nucleus
Synthesis of RNAs and Proteins • 4 promoters and 2 enhancers, liver-enriched transcription factor at least 5 RNA • 5’-cap 3’- polyA (TATAAA) • Common 3’ end seem to be unspliced • X, preS1/S2/S, C/Pol
X Protein • Dispensable for infection, viral replication efficient infection in vivo, potential oncogenicity • The function of X cloning a natural viral infection is not known • Dispensable for viral DNA synthesis in infected tissue culture cells • Activate the transcription of host genes (MHC, C-myc)and viral genes • Stabilizes viral RNAs, not a DNA binding protein • Clear that X-defective virus is unable to initiate infection in vivo
S Protein • 2.1 Kb, does not contain a sharply defined 5’ end, well transcribed in a variety of different cell lines • LHBS - 2.4Kb, transcribe in a tissue-dependent manner • HNF1 – liver specific factor is essential for virus secretion • Three s-glycosylated, type II transmembrane proteins, multimer by disulfide • Dane particle – L and M (equal amount, 30%) • Filamentous and spherical particles – 100-fold excess (~100g/ml) • Ab to S protein is enough • preS1 domain – viral receptor • preS2 – not essential for virus infection
Topological Models of the 3 HBs Proteins B A • Structure of HBs • Structure of MHBs • Structure of LHBs C
Core Protein • One core/e promoter, tissue dependent • 30bp longer – preC sequence • preC – hydrophobic -helix (signal) • Arginine center (C-termus) - bind NA • Maybe involved in 1st and 2nd DNA synthesis translation initiation of pol - internal entry or leaky scanning • Core particle – 36nm
Polymerase • TP, spacer, pol, RNase H • Conserved motif – A~E (YMDD) • TP – critical role in the packaging of pg RNA • Cellular factor – HSP90, HSP70, HSP60
Assembly of the Nucleocapsid • Assembled spontaneously nonspecific incapsidation of RNA • C-terminal is not sufficient to mediate the specific encapsidation • Polymerase is required for specific RNA packing • -sequence, cellular factor -RNA + pol conformation change -RNA + pol core • Protein kinase C activity
Re-entry of Mature Core Particle • New infection • Re-entry : amplification of cccDNA up to 50 copies : mutant DHBV(defective for S protein) 50 fold over • Regulation via envelopment
Envelopment and Secretion of HBV • S proteins : rough ER • Virion formation depends on viral DNA synthesis • Protein maybe phosphorylation of the DHBV core protein • Binding of cores to the N-terminal of S budding of the core particle across the ER membrane • Glycosylation of Asparagine of S blood stream
Entry into the Host Main routes of infection are perinatal, blood and sexual Transmission. Current greatest source of new infections is vertical transmission from infected mothers to newborn. In western hemisphere, risk of vertical transmission is 10%. In Asia, the rate of vertical transmission is almost 90%. Modern screening technologies have dramatically decreased The risk of HBV transmission by blood transfusion, but some HBV variants escape serologic assay. Horizontal transmision can be observed via close bodily. The high rate of chronicity in parts of Africa is attributed to horizontal spread between young children.
Transient infection -1-6 months -e antigen : neonatal tolerogen -decrease hepatocyte half life -> hepatocyte replacement for a short period Chronic infection -high level HBsAg -10-15% of chronic infection -> Cirrhosis, liver cancer -treatment : lamivudine ( inhibition of p protein ) after 1 year, lamivudine resistance virus : a interferon
Sites of HBV Infection Virtually all hepatocytes are infected, glomular infection in the kidney is detectable and a small amount of infection in exocrine cells of the pancreas.
Serological Profiles of HBV Infections Diagnosis of acute hepatitis B. Acute HBV infection is demonstrated by detection of HBsAG and anti-HBc IgM antibody. Diagnosis of chronic hepatitis B. Chronic HBV infection is demonstrated by persistence of serum HBsAG and serum HBeAg and HBV DNA.
Diagnosis of chronic hepatitis B to inactive carrier state. Hbe seroconversion is associated with the loss of serum HBeAg, appearance of anti-HBeAb, decline of serum HBV DNA levels and normalization of ALT levels. Diagnosis of HBeAg negative chronic hepatitis.
Liver Cancer • Direct model • - X protein ( Transgenic mouse ) - Truncated M -> induce integration to host chormosome • (rarely) - Enhancer II -> activation N-myc ( only WHV, not HBV ) - p53 mutation ( 249 codon G->T ) of 80-90% HCC patient • : multiple step - Activation of insulin-like growth factor(IGFII) ( only WHV ) • Woodchucks and ground squirrels chronically infected with WHV or ground squirrel hepatitis virus develop HCC. • Indirect model • - HBV infection ( L protein overexpression )-> liver cell injury
Chronic viral hepatitis showing • inflamed cells and fibrous tissue. A B. Acute viral hepatitis showing disruption of lobular architecture, inflamed cells, and hepato- cellular apoptosis. B C. Cirrhosis resulting from chronic viral hepatitis. C
Integration of HBV Genome HBV DNA sequences have been found in HCC tumor samples and in hepatocytes of patients with chronic hepatitis. There have also been evidence that integration occurs in acute infection. HBV DNA integration is a dynamic process. It has been observed that long term chronic inflammation induces rearrangement of integrated viral DNA. Because integrated HBV sequences can induce chromosomal deletions at the site of integration as well as the observation that transpositions of HBV suequences, HBV can cause chromosomal instability. Furthermore, the integration of the HBV DNA can activate proto-oncogenes and change the regulation or disrupt genes involved with cell proliferation and viability.
Role of HBx in Liver Cancer HBx sequences have been found in large proportions in tumor cells. HBx is expressed at low levels during acute and chronic hepatitis. HBx protein and RNA has been detected in the absence of viral Replication in HCC tumor cells. Mouse studies have shown that HBx induces HCC by sensitizing animals To chemical carcinogens as well as influencing expression of proto-oncogenes. HBx has been found to be able to regulate many different pathways including those involved in DNA cell cycle and apoptosis.
Translational Basis of Some of HBx Functions Inhibition of cell death HBx modulates activity of serine protease hepsin and upregulates survivin providing anti-apoptotic functions. HBx expression stabilizes hypoxia induced factor, Hif-1. HBx can activate Wnt/beta-catenin signaling. HBx activates matrix metallo- proteinase 3 and 9. Angiogenesis Cellular migration
Role of Hepatitis B Virus Spliced protein (HBSP) HBSP has been found in liver biopsy from patients with active chronic hepatitis. Detectable in serum of approx 45% of patients with chronic hepatitis. HBSP can induce apoptosis and regulate TGF-beta signaling. There is strong correlation of HBSP fibrosis.