OPTICAL ISOMERISM
R W Grime Ripon Grammar School. OPTICAL ISOMERISM. Molecular mirror images. Optical isomerism and chirality. Enantiomers and racemates. Self-Test: recognising optical isomers from structural formulae. Polarimetry: recognising optical isomers by experiment.

OPTICAL ISOMERISM
E N D
Presentation Transcript
R W Grime Ripon Grammar School OPTICAL ISOMERISM Molecular mirror images Optical isomerism and chirality Enantiomers and racemates Self-Test: recognising optical isomers from structural formulae Polarimetry: recognising optical isomers by experiment Examples of optical isomers and their importance thalidomide carvone limonene
All molecules have a mirror image – but for most molecules it is the same molecule.
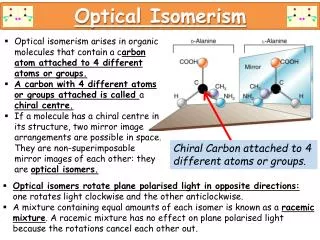
For some molecules the mirror image is a different molecule (the mirror image is non-superimposable).
Left and right hands are an example of non-superimposable mirror images.
This usually happens when a molecule contains a C atom with four different groups attached (chiral / asymmetric C). • Such molecules are said to be chiral or optically active.
The optical isomers are called enantiomers. • These are distinguished by +/-, D/L or more correctly R/S. • A 50/50 mixture of the two enantiomers is called a racemic mixture or a racemate.
TASKSome of the following molecules are optically active. For each one, click its name below and decide whether it is optically active or not. Click again to see if you are correct. a) propan-2-ol e) butanone b) 2-chlorobutane f) 2-methylbutanoic acid c) 1-chlorobutane g) butan-2-ol d) 3-methylhexane h) 1-chloro-3-methylpentane
propan-2-ol NOT OPTICALLY ACTIVE Click here to go back to the optical isomerism task
OPTICALLY ACTIVE 2-chlorobutane Click here to go back to the optical isomerism task
1-chlorobutane NOT OPTICALLY ACTIVE Click here to go back to the optical isomerism task
OPTICALLY ACTIVE 3-methylhexane Click here to go back to the optical isomerism task
butanone NOT OPTICALLY ACTIVE Click here to go back to the optical isomerism task
propan-2-ol NOT OPTICALLY ACTIVE Click here to go back to the optical isomerism task
OPTICALLY ACTIVE 2-methylbutanoic acid Click here to go back to the optical isomerism task
OPTICALLY ACTIVE butan-2-ol Click here to go back to the optical isomerism task
OPTICALLY ACTIVE 1-chloro-3-methylpentane Click here to go back to the optical isomerism task
Molecules that are optical isomers are called enantiomers. • Enantiomers have identical chemical and physical properties, except: • Their effect on plane polarised light; • Their reaction with other chiral molecules
The wave vibrations are perpendicular to the direction of travel of the wave.
POLARIMETERS can be used to analyse the effect optical isomers have on plane polarised light: Heriot Watt University has a web page with an interactive tutorial and self-test questions about this topic: http://scholar.hw.ac.uk/site/chemistry/activity5.asp?outline
Chiral molecules often react differently with other chiral molecules. • This is like the idea that a right hand does not fit a left handed glove – the molecule must be the correct shape to fit the molecule it is reacting with. • Many natural molecules are chiral and most natural reactions are affected by optical isomerism.
For example, most amino acids (and so proteins) are chiral, along with many other molecules. • In nature, only one optical isomer occurs (e.g. all natural amino acids are rotate polarised light to the left).
Many drugs are optically active, with one enantiomer only having the beneficial effect. • In the case of some drugs, the other enantiomer can even be harmful, e.g. thalidomide.
In the 1960’s thalidomide was given to pregnant women to reduce the effects of morning sickness. • This led to many disabilities in babies and early deaths in many cases. The photographs are both from ‘Molecule of the Month’ at Bristol University: http://www.chm.bris.ac.uk/motm/thalidomide/start.html
S thalidomide (effective drug) The body racemises each enantiomer, so even pure S is dangerous as it converts to R in the body. R thalidomide (dangerous drug)
Thalidomide was banned worldwide when the effects were discovered. • However, it is starting to be used again to treat leprosy and HIV. • Its use is restricted though and patients have to have a pregnancy test first (women!) and use two forms of contraception (if sexually active).
S carvone (caraway seed) R carvone (spearmint) Caraway Seed has a warm, pungent, slightly bitter flavour with aniseed overtones.
S limonene (lemons) R limonene (oranges)