Isomerism
Isomerism. Extended materials Jelena Marinković. First class: Isomerism-revision.

Isomerism
E N D
Presentation Transcript
Isomerism Extended materials JelenaMarinković
First class: Isomerism-revision The existence of chemical compounds that have the same molecular formulae but different molecular structures or different arrangements of atoms in space is called isomerism. In structural isomerism the molecules have different types of compound or they make simply differ in the position of the functional group in the molecule. Structural isomerism generally have different physical and chemical properties. In stereoisomerism , the isomers have the same formula and functional groups, but differ in the arrangement of groups in space. Optical isomerism is one form of this. Another type is cis-trans isomerism, in which the isomers have different positions of groups with respect to a double bond or ring or central atom.
The relationship of different types of isomers Isomers Structural Isomers StereoisomersDifferent bonding Different shapes Students should explain different types of isomerism, and how that influence to the physical and chemical properties of compounds.
Working in the groups: • Students are divided in four groups. • First group has a task to make different structural isomers of a molecular formula C8H18. • Second group has a task to make different cis-trans isomers of octen. • Third group has a task to make different isomers of dimetilcyclo pentane. • Fourth group has a task to make different optical isomers of buthanol, penthanol and hexanol. • At the end of the class all the groups have to present their results and discuss about the the results.
Strategies: • Collaborative work amongst students and between students and teacher. • A participatory approach to class, with students taking responsibility for contributing. • Diversity of teaching methods, resources and aids.
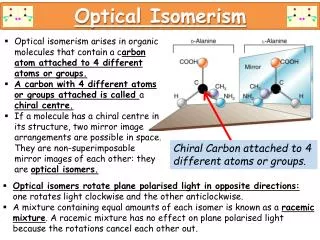
Second class: Detailed explanation of optical isomerism • If a carbon atom has four different groups attached to it then there are two different ways in which these groups can be arranged around this carbon atom, which is known as a chiral or asymmetriccarbon atom. The two forms of the molecule, which are known as enantiomers, are mirror images of each other that cannot be superimposed (like a pair of gloves),. This is illustrated below using 2-aminopropanoic acid (alanine) as an example.
Because these molecules are so similar, there is very little difference in their physical and chemical properties. In fact the only difference is that they have differing effects on polarised light, one isomer rotating the plane of polarisation clockwise, the other anticlockwise . Biological systems are much more sensitive to the shape of the molecule and so the different enantiomers usually have different biological effects, for example, one isomer of aspartame tastes sweet, but the other enantiomer tastes bitter.
If a molecule has four different groups attached to a single carbon atom, then the compound can exist as a pair of enantiomers. The only difference in the properties of these compounds is in their interaction with plane polarised light. Plane polarised light can be considered to be light in which the oscillation of the wave is restricted to one plane, say the vertical. This can be achieved by passing the light through a polarising filter. If the light is now passed through a second polarising filter orientated in the same direction (e.g. vertical) then there is virtually 100% transmission . If the second polarising filter has its axis at right angles to the first (e.g. horizontal) then no light will pass. A pure enantiomer placed between the two filters will rotate the plane of polarisation in one direction (say clockwise) so that maximum transmission is no longer when the second filter is aligned with the first one. The second enantiomer will rotate the plane of polarisation by exactly the same amount but in the opposite direction (anticlockwise). Substances that affect polarised light in this way are said to be optically active. An instrument containing two polarising filters that can be rotated relative to each other, so as to allow the angle between their orientations to be measured is called a polarimeter.
The other enantiomer would rotate it an equal amount but anticlockwise. Apart from this the properties of enantiomers are identical. Chemically the behaviour of the enantiomers is identical unless the reaction also involves a pure enantiomer. • Chemical reactions that produce an asymmetric carbon atom in a molecule give rise to a mixture containing exactly equal amounts of the two enantiomers. Such a mixture is known as a racemicmixture. The effects of the two enantiomers in a racemic mixture cancel each other out and so it is not optically active. In contrast almost all natural products, produced by enzyme catalysed biochemical processes, result in just one pure enantiomer and hence produce optically active material. Pure turpentine (produced from pine tree resin) can, for example, be differentiated from white spirit (a substitute produced by the chemical industry) because turpentine is optically active and will rotate the plane of polarised light, whereas white spirit will not.
Strategies: • Collaborative work amongst students and between students and teacher. • Diversity of teaching methods, resources and aids.
Third class: Monosaccharides and disaccharides • The term carbohydrate was originally used to describe compounds that were literally "hydrates of carbon" because they had the empirical formula CH2O. In recent years, carbohydrates have been classified on the basis of their structures, not their formulas. They are now defined as polyhydroxyaldehydes and ketones. Among the compounds that belong to this family are cellulose, starch, glycogen, and most sugars. • There are three classes of carbohydrates: monosaccharides, disaccharides, and polysaccharides. The monosaccharides are white, crystalline solids that contain a single aldehyde or ketone functional group. They are subdivided into two classes aldoses and ketoses on the basis of whether they are aldehydes or ketones. They are also classified as a triose, tetrose, pentose, hexose, or heptose on the basis of whether they contain three, four, five, six, or seven carbon atoms. • With only one exception, the monosaccharides are optically active compounds. Although both D and L isomers are possible, most of the monosaccharides found in nature are in the D configuration.
Homework: • Students should find different sources of information about sugars and isomerism • Results: • http://en.wikipedia.org/wiki/Monosaccharide • http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/C/Carbohydrates.html • http://chemistry2.csudh.edu/rpendarvis/monosacch.html • http://www.chemguide.co.uk/basicorg/isomerism/optical.html • http://en.wikipedia.org/wiki/Isomer
Strategies: • Collaborative work amongst students and between students and teacher. • Diversity of teaching methods, resources and aids. • Subject material with “real life application” and connections to the world outside the classroom.
Fourth class: Determining the monosaccharides and stereoisomers • Students are in four groups. Each group gets cards with structural formulae of monosaccharides and their names at separated cards. They have to link structures with names, and to determine α and β stereoisomers. After that, they have to make disaccharides making different bonds between those monosaccharides . • At the end of the class groups have to exchange their papers and make corrections if they are necessary.
Strategies: • Collaborative work amongst students and between students and teacher. • Diversity of teaching methods, resources and aids. • Practical work in tandem with theory.
Conclusion • Student were very interested in this topic. They prefer working within a group. They are looking forward going to the Science University, to do laboratory investigations. • I expect that test results are going to be better than usually, because I notice that they are a little bit more involved in learning. • The most important is that students realized how small number of monosaccharides, making different isomers and different bond, produce a large number of di- and polisaccharides.