Understanding Enzymes: Nature’s Biological Catalysts
200 likes | 321 Views
Enzymes are specialized proteins that act as biological catalysts, significantly speeding up chemical reactions in living organisms. They lower the activation energy required for reactions, allowing vital processes to occur rapidly, which is essential for life. Each enzyme is specific to a particular substrate, functioning like a 'key' for a 'lock', ensuring precise biochemical reactions. Various factors influence enzyme activity, including temperature and pH, and enzymes are crucial in industries such as food production and detergents. Their specificity also enables the development of inhibitors for drugs and poisons.

Understanding Enzymes: Nature’s Biological Catalysts
E N D
Presentation Transcript
KeyWords • Catalyst – A chemical substance that speeds up a reaction. It is not used up in the reaction. • Protein - Organic molecules made up of amino acids. They have vital roles in all cells as building materials e.g keratin, insulin, haemoglobin, collagen. • Enzyme – Are biological molecules that catalyse many different reactions. Most enzymes are proteins. e.gamylase, lipase, protease
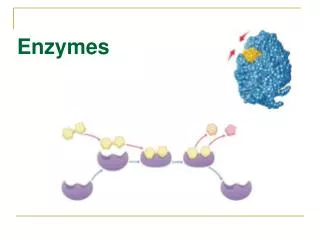
Why Enzymes? • Enzymes allow chemical reactions in the body to happen quickly enough to keep us alive. • The enzyme acts like a ‘key’ that will only fit a specific ‘lock’. The lock is the reactants or substrates (starting substances). The enzyme brings the substrates together and changes them into different molecules called the products.
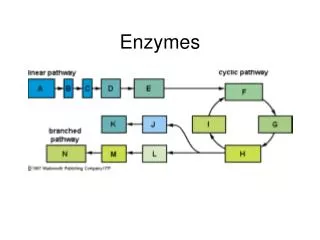
Activation Energy • All catalysts work by lowering the activation energy. This is the starting energy required to begin the reaction. • Enzymes therefore lower the activation energy required to start the reaction, this allows the reaction to happen much faster BUT remember only the enzyme that fitsthe substrates will work to speed up the reaction! • This allows specific reactions to happen when we need them (a series of enzyme reactions is called a metabolic pathway – almost like pick a path.)
Catalyst or Enzyme? • Both catalysts and enzymes lower the activation energy of the reaction allowing the reaction to happen millions of times faster than it would usually occur • Both are NOT used up in the reaction, they just speed it up and allow it to happen • HOWEVER, enzymes are much more specific than catalysts • Enzymes are known to catalyse 4,000 biochemical reactions!
Drugs? • Due to the specific lock and key fit of an enzyme with a substrate Scientists have been able to design inhibitors that will fit into the enzyme and block it from joining the substrate. • This is good if you want to stop the reaction from occuring, BUT some poisons will also do this and stop vital enzymes from starting reactions that we need in order to survive • Cyanide stops the enzyme that allows energy production in the mitochondria of our cells. This causes death and was used in the US gas chambers as a method of execution
More inhibitors • Some inhibitors bind to the active site – this is the site where the substrates should bind • Some inhibitors do not bind to the active site, but once they are bound to the enzyme they change the shape of it so that the substrates no longer fit into the active site • Some inhibitors do not bind to the active site, AND do not change the shape of the active site, but by being bound to the enzyme they prevent the products from being produced even though the substrates are bound!
Controlling Enzymes • Like microbes, enzymes need certain conditions to optimize their efficiency. These are: • Temperature • pH • Activators • Inhibitors
Temperature • Proteins change shape as temperatures change. Because so much of an enzyme's activity is based on its shape, temperature changes can mess up the process and the enzyme won't work. This is why when our body’s temperature changes from 37o by only a couple of degrees we are at risk of DEATH
pH • In the same way temperature changes the shape of proteins, the acidity of the environment does the same thing. Remember that the pH is a measure of acidity? An increased acidity near an enzyme can cause its shape to change. The enzyme could unravel and become totally ineffective.
Activators • Sometimes you need an enzyme to work faster and your body creates an activator. Other times you might eat something that acts as an activator. Activators make enzymes work harder and faster. If you're running in a race and you need more energy, get those enzymes to work!
Inhibitors • These are the opposite of activators. Inhibitors either slow down or stop the activity of an enzyme. They often bond to the protein, changing the overall shape of the enzyme. Remember, when the shape changes, the enzyme will not work the same way. A nasty example of an inhibitor is snake venom or maybe nerve gas from World War I.
Enzyme technology • Commercial enzymes are obtained from three main sources: Plant, Animal and Micro-organisms • Enzymes are used for beer production. The starch-digesting enzyme amylase changes the barley starch into maltose sugar (which the yeast can then ferment) • The enzyme Papain from papaya is used in meat tenderisation
Enzyme technology cont. • The rennet used for curdling milk is traditionally extracted from the stomachs of unweaned calves • Enzymes such as amylases and cellulases and proteases are used in biological detergents as they break down protein stains (such as egg, blood and milk) and are insoluble in water and cling tightly to fabric