Molarity
140 likes | 698 Views
Molarity. Concentration Based on Mass. Concentration - amount of solute dissolved in a given amount of solution Concentration of a solution has an effect on Physical properties Melting and boiling points Chemical properties Solution reactivity. Moles and Equivalents.

Molarity
E N D
Presentation Transcript
Concentration Based on Mass • Concentration - amount of solute dissolved in a given amount of solution • Concentration of a solution has an effect on • Physical properties • Melting and boiling points • Chemical properties • Solution reactivity
Moles and Equivalents • Chemical equations represent the relative number of moles of reactants producing products • Many chemical reactions occur in solution where it is most useful to represent concentrations on a molar basis
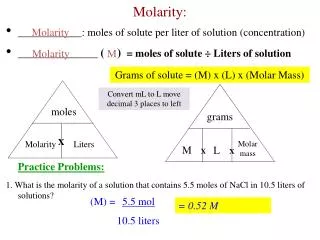
Molarity • The most common mole-based concentration unit is molarity • Molarity • Symbolized M • Defined as the number of moles of solute per liter of solution
Molarity 2M HCl What does this mean?
Molarity Calculations MASS IN GRAMS MOLES NUMBER OF PARTICLES LITERS OF SOLUTION 6.02 1023 (particles/mol) molar mass (g/mol) Molarity (mol/L)
Calculating Molarity from Moles • Calculate the molarity of 2.0 L of solution containing 5.0 mol NaOH • Use the equation • Substitute into the equation: MNaOH = 5.0 mol solute 2.0 L solution = 2.5 M
Molarity Calculations How many grams of NaCl are required to make 0.500L of 0.25M NaCl? 0.500 L 0.25 mol 1 L 58.44 g 1 mol = 7.3 g NaCl
Molarity Calculations Find the molarity of a 250 mL solution containing 10.0 g of NaF. M = 10.0 g 1 mol 41.99 g = 0.238 mol NaF 0.238 mol 0.25 L = 0.95M NaF
Molar Volume of a Gas • Molar volume- the volume occupied by 1 mol of any gas • STP – Standard Temperature and Pressure • T = 273 K (or 0oC) • P = 1 atm • At STP the molar volume of any gas is 22.4 L • Vgas = molesgas x (22.4L/1 mol)
Molar Mass and Density • A gas’s density will determine if it will sink or float in the air • Molar mass = density x molar volume • At STP