Molarity
Molarity. By: Gurpreet Dulay & Kiranjit Kandola. Some definitions. The CONCENTRATION of a substance in solution is the amount of the substance which exists in a given volume of the solution

Molarity
E N D
Presentation Transcript
Molarity By: Gurpreet Dulay & Kiranjit Kandola
Some definitions • The CONCENTRATION of a substance in solution is the amount of the substance which exists in a given volume of the solution • A CONCENTRATED solution has a relatively high concentration (large amount of substance dissolved in a solution) • A DILUTE solution has a relatively low concentration (very little substance is dissolved in the solution)
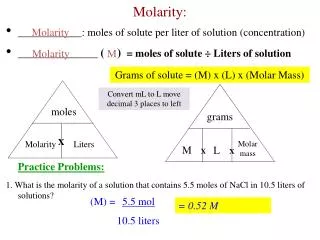
What is Molarity? • The MOLAR CONCENTRATION of a substance in a solution that is the number of moles of the substance contained in 1 L of solution • Molar concentration is also known as MOLARITY • The unit symbol for “mol/L” is “M” • 1 mole is 6.02 x 10²³
How to find molar concentration Molar concentration = Moles Volume or : c = n v Where: c = molar concentration, in mol/Ln = number of molesv = volume, in liters
How to Prepare a Standard Solution • Weigh 0.58 NaCl • Add NaCl to 80mL of water • Stir solution until salt is dissolved • Pour solution into 100 mL grad. cylinder • Bring the solution up to 100 mL
Calculations:Moles, Volume, and Molarity 1) How many moles of AlCl3 are contained in 350.0 mL of 0.250M AlCl3? moles AlCl3 = 0.250 mol x 0.3500 L = 0.0875 molL 2) What volume of 2.40 M HCl can be made from 100.0 g of HCl? moles of HCl = 100.0g x 1 mol = 2.74 mol 36.5 g 2.74 mol = 1.14 L 2.40 mol/L 3) What is the molarity of the CaCl2 in a solution made by dissolving and diluting 15.00 g of CaCl2 x 6H2O [CaCl2]=[CaCl2 x 6H2O] = 15.00g x 1 mol = 0.1369 M 0.5000 L 219.1g
Dilution Calculations • The formula for dilution calculations is: MI VI = MF VF Where MI isthe initial molarity VIis the initial volume MFis the final diluted molarity VFis the final volume
Dilution calculation example: A chemist starts with 50.0 mL of a 0.40 M NaCl solution and dilutes it to 1000 mL of water. What is the concentration of NaCl in the new solution? 50.0 mL of 0.40 M NaCl 1000. mL water
STEPS: You multiply the two intials together: • (0.40 M) (50.0 mL) = (MF) (1000. mL) Then you use basic algebra to isolatethe MF • (0.40 M) (50.0 mL)(1000. mL) = (MF) Final answer: • 0.020 M = MF