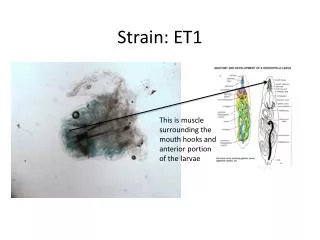
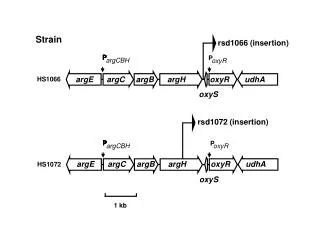
Strain
Assessment of key parameters to achieve bacteria high transformability in E. coli and A. tumefaciens Zach Regelin 1 , Suzette Smedes 2 , Kan Wang 3 , Diane Luth 3 , Ksenija Markovic 3 , Helene Eckert 3 , François Torney 3

Strain
E N D
Presentation Transcript
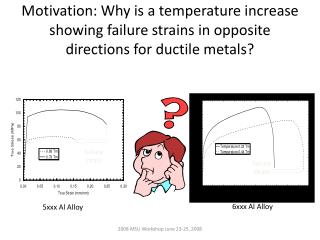
Assessment of key parameters to achieve bacteria high transformability in E. coli and A. tumefaciens Zach Regelin1, Suzette Smedes2, Kan Wang3, Diane Luth3, Ksenija Markovic3, Helene Eckert3,François Torney3 1University of Northern Iowa, 2 Melcher-Dallas Community School District, 3 Dept. of Agronomy, Iowa State University June 13, 2006 – August 2, 2006 Project Description Monitoring the Growth Curve Using E. coli strains Dh5 and ccdBR, it was determined that the strain, the media type, and the amount of inoculum had a great effect on the growth but the source of the inoculum had little effect. (data not shown) Electroporation is a genetic transformation process in which a strong electric field is used to introduce DNA into cells. Bacterial transformation is important for molecular cloning, gene construct selection, and plasmid production and storage. To optimize the electroporation process, several key parameters were investigated, including: the strain of the bacteria, the inoculum source, and the media type. Bacteria were grown under different conditions and their growth was monitored. Bacteria grown to different phases of growth under various conditions were used for electroporation and transformation efficiencies were determined. Figure 2 For A. tumefaciens, the strain and the media type had a great effect on the growth and the source of the inoculum had a great effect for one strain’s growth but little effect for the other’s (Figure 3). Figure 3 Electroporation E. coli in LB E. coli in 2xTY • tumefaciens • in YEP • CcdBR showed higher efficiencies than Dh5. • The higher OD cultures had higher efficiencies. • The media did not have a significant effect. • The standard deviations are too large for these results to be conclusive. Growth Curve 0.6 Species 0.5 Strain Dh5a Plate 0.4 Inoculum ccdBR Plate 0.3 OD Dh5a glycerol Medium 0.2 ccdBR glycerol 0.1 Species 0 0 50 100 150 Strain Time (min) Inoculum Medium Figure 4 Results and Discussion Materials and Methods The growth curves for different E. coli and A. tumefaciens strains were determined by measuring optical densities (ODs) and plating diluted culture samples. Growth curves were determined for different inoculums and different media (Figure 1). Electrocompotent cells were prepared by a series of washing and pelleting in 10% glycerol solution, then flash-frozen in liquid nitrogen and stored at -80ºC. To genetically transform the cells by electroporation, they were mixed with plasmids and a 2.5 kV current was passed through the mix for 5.9 ms. Cultures were plated on selective media to determine transformation efficiencies. Figure 1 Acknowledgement I would like to thank the National Science Foundation REU program for funding this project.