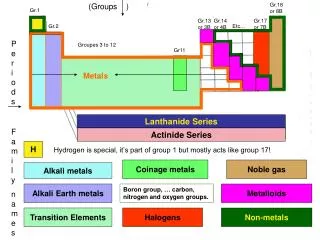
Alkali Metals
Alkali Metals. Aga Smith. Basic Information. The Alkali Metals are the Chemical Metals from Group 1 in the periodic table. The 6 elements from the Alkali Metals group are Lithium (Li), Sodium (Na), Potassium (K), Rubidium ( Rb ), Caesium (Cs), Francium (Fr).

Alkali Metals
E N D
Presentation Transcript
AlkaliMetals Aga Smith
Basic Information • The Alkali Metals are the Chemical Metals from Group 1 in the periodic table. • The 6 elements from the Alkali Metals group are Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Caesium (Cs), Francium (Fr). • Hydrogen (H) is also in group 1 aswell as the Alkali Metals but it has very few things in common with them so it is not concidered an Alkali Metal. • They are highly reactive. • Never found in their element form anywhere in nature. • Usually stored in mineral oil or kerosene (paraffin oil). • They have low melting points • They have low densities • All of the elements from the Alkali Metals group are soft metals.
What Do They Look Like? Most of the time they’re silver coloured An exception is caesium because it has a golden tint.
All the alkali metals react intensively with halogens to form ionic salts. * Ionic salts- usually the result of neutralization from the reaction of an acid and a base. Salts are neutral ( without a charge) composed of cations and anions ( positively and negatively charged ions).
All react with water to make strongly alkaline hydroxides. ( Alkalinehydroxides- class of chemical compounds which is made up of alkali metal cation and hydroxide anion.) The reactivity increases down the group.
All of the atoms in alkali metals have one electron in their outer electron shells. To become stable they have to give off one electron to elements with seven electrons in their outer shell making it eight, also stable. Then they become single charged positive ions (cations). • Melting point decreases as we go down the group • Theatomicweightincreases as we go down the group • Boiling point decreases as we go down the group • Densityincreases as we go down the group
Electron Configuration Lithium Li- 1s2, 2s1 Sodium Na- 1s2, 2s2, 2p6, 3s1 Potassium K- 1s2, 2s2, 2p6, 3s2, 3p6, 4s1 Rubidium Rb- 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6, 5s1 Caesium Cs- 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6, 5s1
Vigorous reactions with water Alkali metal + water → Alkali metal hydroxide + hydrogen gas
Lithium -Soft silver-white metal -highly reactive and flammable -stored in mineral oil -focal lenses, rocket propellants, oxygen candles, glasses, ceramics, used to absorb carbon dioxide in space vehicles -2 stableisotopes Sodium -One stable isotope - Table salt, baking soda, soap, drain cleaners, food preservative,
Potassium -Preservative for beer and wine, medical purposes, fireworks, dies, gunpowder, soap, glass -24 isotopes Rubidium -Glasses, ceramics, fireworks, ion engines for space vehicles, -24 isotopes
Caesium -Discovered by Robert Bunsen and Gustav Kirchhoff in 1860 in Germany. -It’s Latin name ‘caesium’ means sky blue. It is called the way it is because of the blue emissions it has. -To get a puresample of caesium, 44,000 litres of waterhad to be evaporated. -It was discovered spectroscopically -Helps treat many types of cancer -39 isotopes Francium -Discovered in 1939 by Marguerite Perev while analyzing actinium’s decay sequence. - The name was in honor of her country, France. At first it was supposed to be called actinium K. -She determined this element’s radio-chemical and nuclear properties. -Most unstable element that exists. -Thereisverylittlefranciuminnature so scientistsmust make it to be able to studyit.
Similarities of elements in the Alkali Metals group Similarities: -soft -silvery colour -too reactive to be found in nature -one electron in outer shell that is easily given off -react violently with water and give off flammable hydrogen
Bibliography • http://www.carondelet.pvt.k12.ca.us/Family/Science/Alkali%20Metals/ • http://nautilus.fis.uc.pt/st2.5/scenes-e/elem/e08710.html • http://en.wikipedia.org/wiki/Alkali_metals • http://en.wikipedia.org/wiki/Lithium • http://en.wikipedia.org/wiki/Sodium • http://en.wikipedia.org/wiki/Potassium • http://en.wikipedia.org/wiki/Rubidium • http://en.wikipedia.org/wiki/Caesium • http://en.wikipedia.org/wiki/Francium • http://www.lenntech.com/periodic/elements/li.htm