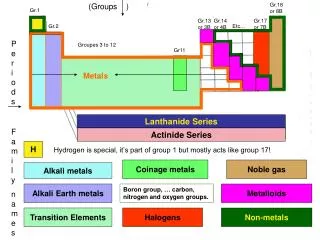
Alkali Metals
Alkali Metals. Lucas Petocz Period 1 October 21 st , 2011. WHAT ELEMENTS ARE IN ALKALI METALS/CHEMICAL PROPERTIES OF ALKALI METALS. The elements in Alkali Metals are Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium.

Alkali Metals
E N D
Presentation Transcript
Alkali Metals Lucas Petocz Period 1 October 21st, 2011
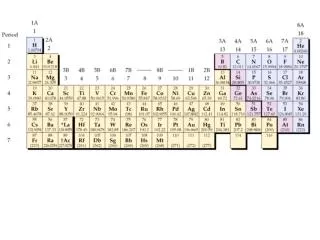
WHAT ELEMENTS ARE IN ALKALI METALS/CHEMICAL PROPERTIES OF ALKALI METALS • The elements in Alkali Metals are Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium. • Chemical properties of Alkali Metals are that they are very reactive, they only have one electron in the outer shell, and they can explode if exposed to water.
Lithium • Its name comes from the Greek word lithos, meaning stone. • Has a tendency to lose an electron. • Is used in batteries and medicine.
Sodium • Its name comes from the code name for soda, Na2CO3. • Causes the orange color in street lamps. • Generally a sodium salt of fatty acids.
Potassium • Name comes from the English word potash, and the Arabic word qali. • Was previously used to make soap by mixing it with animal fat. • Originally was confused with sodium.
Rubidium • Name comes from the Latin word, rubidus. • Ignites spontaneously with air and violently with water. • Can only be a liquid at ambient temperatures on a hot day.
Cesium • From the Latin word, caesius, or “sky blue”. • Was the poisonous substance emitted from the Fukushima nuclear disaster. • Used in the hydrogenation of organic compounds.
Francium • Francium is named after the country France. • Can be made artificially by bombarding thorium with protons. • Is vanishingly rare.
Short Video • http://www.youtube.com/watch?v=uixxJtJPVXk