Understanding Diffusion and Osmosis: Membrane Dynamics and Water Potential
This lab focuses on the principles of diffusion and osmosis through selectively permeable membranes. Key concepts include the structure of phospholipid bilayers, the movement of water and solutes, the impact of turgor pressure in plant cells, and the significance of water potential. Students will explore how different concentrations of solute affect water movement via osmosis, calculate solute potential using iCRT, and conduct experiments with agar and potato pieces to observe diffusion rates. Engaging multimedia resources supplement the learning experience.

Understanding Diffusion and Osmosis: Membrane Dynamics and Water Potential
E N D
Presentation Transcript
Diffusion and Osmosis Lab 2 Background
Membranes • Phospholipid bilayer containing embedded proteins • Fatty acids limit the movement of water because of their _______________.
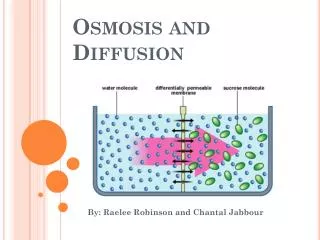
Water • Passes freely through the membrane by osmosis or through aquaporins. • Moves from areas of high potential/high [water] to low potential of low [water]. * In walled cells, osmosis is affected by [solute] & resistance to water movement in the cell by the cell wall (known as Turgor Pressure).
Ions • Move through protein channels
Larger Molecules • Ex. Carbohydrates • Use transport proteins
Diffusion • Simplest form of movement • Solute move from areas of high [solute] to areas of low [solute] • Does not require energy input *ATP and protein carriers called Pumps are required to move solute from areas of low [] to areas of high [].
Diffusion • Molecular weight is inversely related to the rate of diffusion • Non-directional • Increases as kinetic energy increases (for example via increase in temperature) • At equilibrium, diffusion reaches a dynamic equilibrium not static equilibrium.
Solute concentrations • Hypertonic- high [solute]; low water potential • Hypotonic- low [solute]; high water potential • Isotonic- equal water potentials • The above 3 terms are used to describe solutions separated by selectively permeable membranes • These terms are used to describe solute concentration. • How does solute reduce free water in a system? • Which way will water move through a selectively permeable membrane in the above solutions?
Homeostasis • Cells must maintain their internal environments and control solute movement • Why?
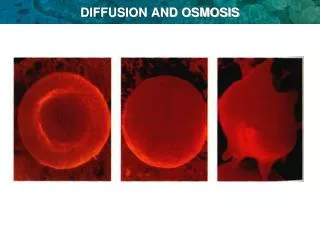
Water Potential • As water moves out of the cell, the cell shrinks or undergoes crenation. • As water moves into the cell, it may eventually burst or lyse. • In walled cells (fungal & plant), the cell wall prevents the cell from bursting as water enters but pressure builds up inside the cell and affects the rate of osmosis.
Water Potential • Measures the tendency of water to move from 1 compartment to another • Is the free energy/mole of water • Represented by the Greek letter psi ____. • = pressure potential (+ve or –ve) + solute potential (aka osmotic potential) *pressure potential = exertion of pressure * Solute potential = [solute]
Calculating Solute Potential • Solute potential ____= -iCRT i=ionization content C= molar concentration R= pressure constant = 0.0831 liter bars/mole-K T= temp in K (273 + degree Celcius) * Bar- a metric measure of pressure and is the same as 1 atmosphere at sea level
Hints • Water potential ____of pure water =0 • Water potential ____of a solution = -ve value • Why? *Summary videos: http://www.youtube.com/watch?v=nDZud2g1RVY http://www.youtube.com/watch?v=Ez1oeY0L0oQ
Practice • Calculate the solute potential of a 0.1M NaCl solution at 25 degree Celcius. If the concentration of NaCl inside the plant cell is 0.15M, which way will the water diffuse if the cell is placed into the 0.1M NaCl solution? • What must turgor pressure equal if there is no net diffusion between the solution and the cell?
Part 1 • Think-pair-share: discuss with your lab partners the answers to the questions under getting started on pgs. S54-S55. • Conduct Step 1 • Conduct Step 2 & answer the questions * Cut 3 square blocks of agar (1cm, 2cm & 3cm) *Don’t forget to record your rate of diffusion.
Part 2 • Think-pair-share: discuss with your lab partners the answers to the questions under getting started on pgs. S56-S57. • Set up your experiment- you should 5 cups, don’t forget to label your cups with what solution is inside tube and what soln is outside • Get initial weight; after 30 mins and after 24hrs.
Dialysis Videos • http://www.youtube.com/watch?v=2Th0PuORsWY&feature=related • http://www.youtube.com/watch?v=DRHKq0piN0M&feature=related
Elodea Video • http://www.youtube.com/watch?v=VK-_YHakvho
Part 3 • Think-pair-share: answer question in Step 1 S58-S59. • Skip Step 2 • Design and Conduct your investigation (use potato pieces of the same size however you may vary the type of potato) • Leave overnight • Answer questions on S59 • Rank solutions from lowest to highest solute concentrations • Rank solutions from lowest to highest water potential.