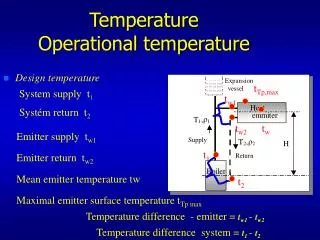
Temperature
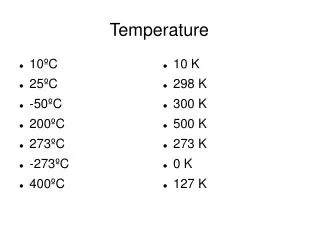
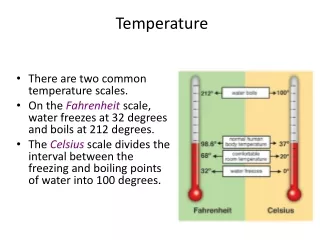
10 K 298 K 300 K 500 K 273 K 0 K 127 K. Temperature. 10 ºC 25ºC -50ºC 200ºC 273ºC -273ºC 400º C. 1 kPa 760 torr 760 mm Hg 101.325 atm 760 torr 1013.25 mm Hg 300 kPa. Pressure. 1 atm 1 atm 760 torr 101.325 kPa 760 kPa 1013.25 mbar 300 torr. 4.67 L 8.4 L. Problems.

Temperature
E N D
Presentation Transcript
10 K 298 K 300 K 500 K 273 K 0 K 127 K Temperature • 10ºC • 25ºC • -50ºC • 200ºC • 273ºC • -273ºC • 400ºC
1 kPa 760 torr 760 mm Hg 101.325 atm 760 torr 1013.25 mm Hg 300 kPa Pressure • 1 atm • 1 atm • 760 torr • 101.325 kPa • 760 kPa • 1013.25 mbar • 300 torr
4.67 L 8.4 L Problems • 2.0 L of gas at 1.5 atm is changed to a pressure of 3.5 atm • 4.2 L of gas at 100ºC is altered to 200ºC
4.67 L 8.4 L Problems • 2.0 L of gas at 1.5 atm is changed to a pressure of 3.5 atm • 4.2 L of gas at 100ºC is altered to 200ºC
18.6 L 3.11 L Problems • 9.3 L of gas at 450 K is changed to a temperature of 900 K • 1.75 L of gas at 225 kPa is altered to 400 kPa
2.25 L 4.31 L Problems • 2.5 L of gas at 50ºC is changed to standard temperature • 1.75 L of gas at 2500 mbar is changed to standard pressure