ISOTOPES
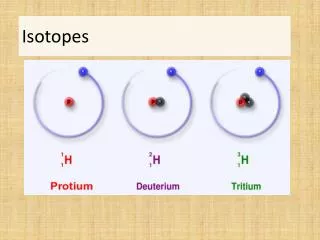
ISOTOPES. Mass Number – total # of protons & neutrons in the nucleus of an isotope Isotope – atoms of the same element that have different masses Isotopes of an element have same # of protons & electrons but different # of neutrons. ISOTOPES. Naming Isotopes Hyphen notation

ISOTOPES
E N D
Presentation Transcript
ISOTOPES • Mass Number – total # of protons & neutrons in the nucleus of an isotope • Isotope – atoms of the same element that have different masses • Isotopes of an element have same # of protons & electrons but different # of neutrons
ISOTOPES • Naming Isotopes • Hyphen notation • Mass number is written with a hyphen after the name of the element Name-mass # • Ex. Hydrogen-3 • Nuclear symbol notation • Superscript indicates mass number • Subscript indicates atomic number • Ex. Uranium-235 would be written as U 235 92
ISOTOPES • The number of neutrons is found by subtracting atomic number from mass number. Ex: mass # – atomic # = # of neutrons 235 – 92 p = 143 n Average Atomic Mass • The weighted average of the atomic masses of the naturally occurring isotopes of an element.
Calculating Avg. Atomic Mass • For each isotope, multiply the mass by the % abundance. Add the answers together to get the average atomic mass. • Ex: Two naturally occurring isotopes of copper: • 69.17% copper-63; mass 62.929 598 amu • 30.83% copper-65; mass 64.927 793 amu • Find the average atomic mass of copper (0.6917)(62.929 598)+(0.3083)(64.927 793)= 63.55 amu