Isotopes
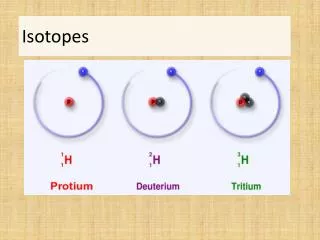
Isotopes. Definition. All atoms of the same element have the same atomic number, but may have different masses. Having the same atomic number, means having the same number of protons (and electrons).

Isotopes
E N D
Presentation Transcript
Definition • All atoms of the same element have the same atomic number, but may have different masses. • Having the same atomic number, means having the same number of protons (and electrons). • The difference in mass is due to the difference in the number of neutrons a specific atom may have. Isotopes
Atomic mass Examples Isotopes • Carbon-12 • Carbon-13 • Carbon-14 Atomic number 12C 13C 14C 35Cl 37Cl • Chlorine-35 • Chlorine-37 17 17 6 6 6
Neutron Formula • Atomic mass – Atomic number = # neutrons Isotopes Examples Atomic mass 12 – 6 = 6 no 12C 14C 6 6 Atomic number 14 – 6 = 8 no
Explanation • So the atomic mass refers to the average mass of all the atoms of an element. Atomic mass calculations Example 100 total atoms of carbon: 99 atoms of carbon-12 1 atom of carbon-13 99 x 12 = 1188 u 1 x 13 = 13 u Total 1201 u Average mass = 1201 u / 100 = 12.01 u
Natural Abundance Atomic mass calculations 98.89 % 1.11 % Example a Steps mass of carbon-12 => 12 x 0.9889 = 11.8668 u mass of carbon-13 => 13 x 0.0111 = 0.1443 u Average mass of naturally occurring Carbon 12.0111 u 12C 13C 6 6
Example b Calculate the atomic mass given the information: Isotope Abundance (%) Mass (u) Cl-35 75 35 Cl-37 25 37 Atomic mass calculations Steps mass of Cl-35 => 35 x 0.75 = 26.25 u mass of Cl-37 => 37 x 0.25 = 9.25 u Average mass of Cl 35.50 u
Example c If 50 % of Bromine atoms have a mass number of 79 and 50 % have a mass number of 81. Calculate the average mass (atomic mass ) of Bromine atoms. Atomic mass calculations Steps 79 x 0.50 = 39.50 u 81 x 0.50 = 40.50 u Average mass of Bromine 80.00 u
Example d If 80 % of Boron atoms have a mass number of 11. The rest have a mass number of 10. What is the atomic mass of Boron? Atomic mass calculations Steps 11 x 0.80 = 8.8 u 10 x 0.20 = 2.0 u Average mass of B 10.8 u
Example e Calculate the atomic mass of Fe. 73 % of the atoms have a mass of 55. 15 % of the atoms have a mass of 56. 12 % of the atoms have a mass of 54. Atomic mass calculations Steps 55 x 0.73 = 40.15 u 56 x 0.15 = 8.40 u 54 x 0.12 = 6.48 u Average mass of Fe 55.03 u