Electronegativity
Electronegativity. Anish Tailor, Jimmy Tang, Costco Bovalis, Jinwoo Oak, Brian Jestice . What is Electronegativity?.

Electronegativity
E N D
Presentation Transcript
Electronegativity Anish Tailor, Jimmy Tang, Costco Bovalis, Jinwoo Oak, Brian Jestice
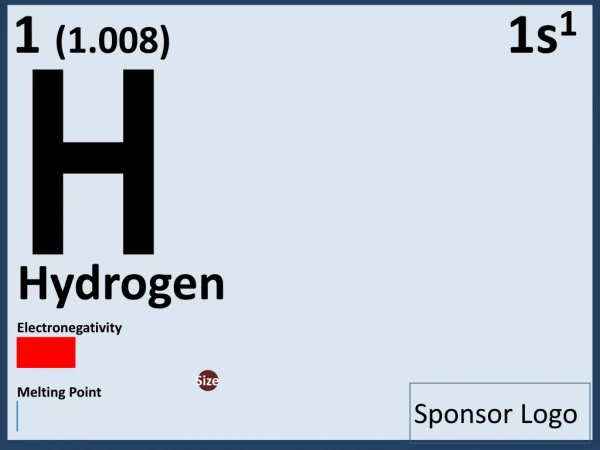
What is Electronegativity? Electronegativity is the measure of attraction between the nucleus and shared electrons of an atom. The more electronegative an atom is, the stronger the attraction between electrons. Electronegativity is measured on a scale from 0 to 4 with 4 being high electronegativity.
This chart is super helpful. High Electronegativity Low Electronegativity
Trend in a row Moving across a row increases electronegativity.
AP Explanation Electronegativity is the tendency of an atom to attract electrons towards itself. Higher atomic number results in more protons in the nucleus, and thus a stronger positive electric charge to attract electrons. In addition, the atom size shrinks from the left to the right of a period.
Trend in a column • Moving down a column decreases electronegativity. Until Cr. • Moving from left to right electronegativity increases.
AP Explanation Increased shielding from an increasing number of electron shells discourages external electrons from being attracted to the atom. Consider Fluorine and Chlorine. Fluorine has only the 1s2 electron shells shielding electrons from the nucleus while Chlorine has 1s2 2s2 2p6 electron shells to shield electrons from the nucleus. Like charges repel, so there is less electronegativity in Chlorine. In each case the pull of the nucleus is +7. However, as we see in the equation F = kqq/r^2, the smaller radius in the Fluorine atom due to less electron shells results in a greater net force.
Units Electronegativity does not have any units, rather, it measured with the Pauling scale. Like the PH scale, the Pauling scale gives a dimensionless quantity ranging from 0.7 to 3.98. Troll Units.
Exceptions and Explanations Although electronegativity increases from left to right, the noble gases are not included because noble gases don't usually bond, though under high pressures Xe and Kr can bond with F.