Ethers
Ethers. Physical Properties Nomenclature Synthesis Reactions. Ethers. are like water, with alkyl groups replacing both of the -H s: H -O- H water R-O- H alcohol R-O-R’ ether. are relatively unreactive. are commonly used as solvents. Physical Properties of Ethers.

Ethers
E N D
Presentation Transcript
Ethers Physical Properties Nomenclature Synthesis Reactions
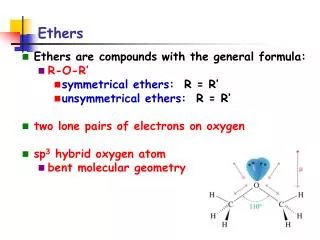
Ethers • are like water, with alkyl groups replacing both of the -Hs: • H-O-H water • R-O-H alcohol • R-O-R’ ether. • are relatively unreactive. • are commonly used as solvents.
Physical Properties of Ethers • The R-O-R’ group is strongly polar. • Ethers have two C-O bonds. • Ethers cannot form internal H bonds. • Their boiling points are lower than those of their alcohol counterparts. • Ethers are better solvents than alcohols for nonpolar compounds.
Physical Properties of Ethers • Ethers can H bond with water and alcohols. • DME (aka glyme), THF, and dioxane are miscible with water.
Physical Properties of Ethers • Ethers are polar, aprotic solvents. • Excellent solvents for strong bases. • Alcohols cannot dissolve bases stronger than RO-. They will instead neutralize the base. • ROH + B- RO- + HB
Physical Properties of Ethers • Ethers solvate cations but not anions. • Ethers make small, hard anions better nucleophiles. • Crown ethers allow inorganic salts to dissolve in nonpolar organic solvents. • THF stabilizes BH3.
Nomenclature of Ethers • Common: alkyl alkyl ether • IUPAC: alkoxy alkane IUPAC: methoxyethane common: methyl ethyl ether IUPAC: methoxybenzene common: methylphenyl ether, anisole
Nomenclature of Ethers • IUPAC: alkoxy alkane • Use the more complex group as the root name. IUPAC: trans-2-methoxycyclohexanol IUPAC: 3-(t-butoxy)-pentane
Nomenclature of Cyclic Ethers • A heterocyclic compound has one or more atoms in the ring that are not C. • The heteroatom is counted as position 1.
Nomenclature of Cyclic Ethers • Epoxides may be named as substituents or as the main group, in which case the word “oxirane” is used. trans-3,4-epoxyhexane trans-2,3-diethyloxirane
Synthesis of Ethers • Williamson ether synthesis (SN2) • alkoxymercuration-demercuration • bimolecular dehydration of alcohols • Epoxides: peroxyacid epoxidation • Epoxides: base-promoted cyclization of halohydrins
Synthesis of Ethers - Williamson Ether Synthesis • SN2 attack of an alkoxide ion on an unhindered 1° alkyl halide or 1° alkyl tosylate. • Elimination competes for 2° halides or 2° tosylates. • Most reliable and versatile of the ether syntheses.
Synthesis of Ethers - Williamson Ether Synthesis redox reaction SN2
Synthesis of Ethers - Williamson Ether Synthesis • Using phenol and dimethyl sulfate, how would you make methyoxybenzene (anisole)?
Synthesis of Ethers - Alkoxymercuration-Demercuration • Forms an ether from an alkene. • Mercurinium ion intermediate…no carbocation, no rearrangement. Markovnikov product anti addition of H - OR
Synthesis of Ethers - Bimolecular Dehydration of Alcohols • After protonation, the 1° alcohol can undergo an SN2 attack by another 1° alcohol molecule to form a symmetric ether. • Competes with alkene formation. • Lower temperatures favor ether formation. 140°C H2SO4
Synthesis of an Epoxide - Reaction of Peroxyacid with an Alkene • Adds an O to the C=C. • Requires a peroxyacid such as MCPBA. • Product is stable if solution is not too acidic. • One-step: no intermediates • No opportunity to change geometry
Synthesis of an Epoxide - Reaction of a Peroxyacid with an Alkene m-chloroperoxybenzoic acid
Synthesis of Ethers - Base-Promoted Cyclization of Halohydrins • alkene + Cl2/water chlorohydrin • Internal SN2 attack in the presence of a base to form the epoxide. SN2 attack
Reactions of Ethers • Cleavage by HBr and HI • auto-oxidation • Epoxides: acid-catalyzed ring opening • Epoxides: base-catalyzed ring opening • Epoxides: ring-opening with Grignard reagents
Reactions of Ethers - Cleavage by HBr and HI • Ethers are unreactive toward bases, but can be protonated by HBr or HI. • Converts a dialkyl ether into two alkyl halides. • R-O-R’ + 2HI RI + R’I + H2O • The nucleophilic substitution may be SN1 or SN2.
Reactions of Ethers - Cleavage by HBr and HI What is the leaving group? SN2 on 1° C SN1 on carbocation, SN2 also possible
Reactions of Ethers - Cleavage by HBr and HI • Converts a phenyl ether into an alkyl halide and a phenol. • sp2 C of phenol cannot undergo SN1 or SN2
Reactions of Ethers - Autooxidation • You should know about this if you ever work in the lab with ethers. • O2 causes the formation of peroxides. R-O-CH2-R’ + excess O2 R-O-O-CH2-R’ + OOH | R-O-CH-R’ can explode when distilled or evaporated
Reactions of Epoxides: Acid-Catalyzed Ring Opening • In water, forms a glycol with anti stereochemistry.
Acid-Catalyzed Opening of Epoxides meso-butan-2,3-diol
Reactions of Epoxides: Orientation of Acid-Catalyzed Ring Opening
Reactions of Epoxides: Acid-Catalyzed Ring Opening • In an alcohol,ring-opening leads to an alkoxy alcohol with anti stereochemistry. • If HBr or HI is the acid, a dihalide is the product. • Rarely useful because the dihalide may be formed directly by the addition of X2 to a C=C.
Reactions of Epoxides: Base-Catalyzed Ring Opening • Requires harsher conditions than the acid-catalyzed ring opening. • SN2 attack of the base on the less-hindered C of the epoxide. • If the base is OH-, the product is a glycol. • If the base is an alkoxide, the product is an alkoxy alcohol.
Reactions of Epoxides: Ring-Opening with Grignard Reagents • The Grignard reagent or organolithium compound is a strong nucleophile, so the attack is like that of the strong base. • With organolithium compounds, the attack is always on the less-hindered C. • With Grignard reagents, there may be a mix of products.
What’s Ahead in Organic II • Aromatic compounds • Ultraviolet (UV) spectroscopy, mass spectroscopy (MS), and conjugated systems • Nuclear magnetic resonance (NMR) spectroscopy • Aldehydes and Ketones • Amines • Carboxylic acids and their derivatives • Additions and Condensations of Enols and Enolates