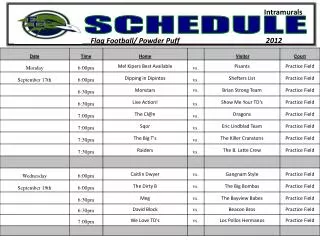
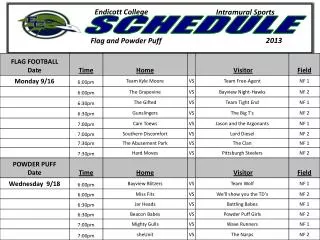
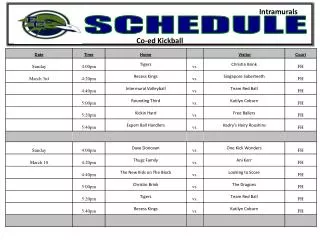
Schedule
Schedule. Schedule. Unilever ’ s Bread & Butter. The Chemistry of Love… . Initial attraction through nonverbal communication is often attributed to phermones. “ Raw lust ” is characterized by high levels of testosterone?

Schedule
E N D
Presentation Transcript
The Chemistry of Love… • Initial attraction through nonverbal communication is often attributed to phermones. • “Raw lust” is characterized by high levels of testosterone? • The sweaty palms and pounding heart of infatuation are caused by higher than normal levels of norepinepherine. • The 'high' of being in love is due to a rush of phenylethylamine and dopamine. • Lasting love confers chemical benefits in the form of stabilized production of serotonin and oxytocin. • Can infidelity be blamed on chemistry? Perhaps in part. Researchers have found that suppression of vasopressin can cause males (voles, anyway) to abandon their love nest and seek new mates.
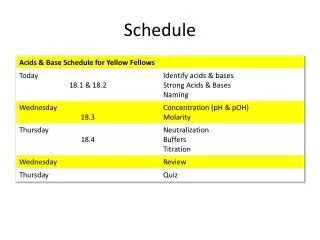
Today’s Objective – 18.1 & 18.2 • To identify acids and bases using the Arrhenius, Brønsted-Lowry and Lewis models. • To identify strong & weak acids and bases. • To name acids & bases. • Tonight’s homework: • read section 18.3 & take notes • mini quiz tomorrow
Content: What is an Acid? • Arrhenius model: A species that contains hydrogen and produces hydrogen ions in an aqueous (water) solution. HX H+ + X- • Real example: HCl H+ + Cl-
Content: What is an Acid? • Brønsted-Lowry model: hydrogen ion donor. Produce conjugate acids and bases HX(aq) + H2O H3O+(aq) + X-(aq) • Real example: HF(aq) + H2O H3O+(aq) + F-(aq)
Content: conjugate acids and bases • conjugate acid: the species produced when a base accepts a hydrogen ion. • conjugate base: the species that results when an acid donates its hydrogen ion. acid base conjugate acid conjugate base
Content: What is a base? • Arrhenius model: species that produces hydroxide ions in an aqueous solution. MOH M+ + OH- • Real examples: NaOH Na+ + OH-
Content: What is a base? • Bronsted-Lowry model: hydrogen ion acceptor. NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) conjugate acid conjugate base base acid
Content: Water – both acid & base H2O + H2O • Known as: amphoteric • Complete the equations. In each, is water an acid or base? • HX + H2O • NH3 + H2O
Summarize: Make Connections • What real world substances are classified as acids and bases? • What ion is likely present in acids?
Today’s Objective – 18.2 & 18.3 • To classify strong acids and bases v. weak acids & bases. • To explain pH and pOH. • Tonight’s homework: • worksheet if you don’t finish it in class.
Content: Strong Acids and Bases • Strong: completely ionizes in aqueous solutions • Strong acid equation • HX (aq) • Strong base equation • MOH
Strong Acids & BasesMemorize Acids Bases Hydroxides of group 1 or 2 metals Ex: NaOH, KOH, Mg(OH)2 • HCl • HBr • HI • HNO3 • H2SO4 • HClO4
Content: Weak acids & bases • Only partially dissociate in water. Reaction does not go to completion, is reversible. • Use double arrow • Weak acid reaction • HB (aq) • Weak base reaction • NH3(aq) + H2O
What does this look like? • http://phet.colorado.edu/en/simulation/acid-base-solutions
Check for Understanding • Classify the following as weak/strong acid/base. • HF • LiOH • HBr • NaOH • NH3
Check for Understanding • What would happen to the following substances in water? Write an equation for each and draw what this would look like • HF -LiOH • NaOH -NH3 • HBr
Classification of acids and Bases • If [H+] > [OH-] = • If [OH-] > [H+] = • If [H+] = [OH-] =
Today’s Objective • To understand pH. • To neutralize an acid using titration.
18.3 Ion Product Constant for Water • Pure water contains equal concentrations of H+ and OH– ions. • The ion production of water • Kw = [H+][OH–] = 1.0x10-14 • a number that doesn’t change, a constant number. • equals the concentration of the H+ and OH– ions.
Ion Product Constant for Water • In an acidic or basic solution, as the [H+] goes up, [OH-] goes down, and vice versa so Kw remains the same.
The pH scale • Scientific notation is cumbersome, so it’s re-expressed using logs. • pH is the negative logarithm of the hydrogen ion concentration of a solution. pH = –log [H+]
Did she just say “log?!” • 103 = 1000 • 10 is the base. • 3 is the exponent. • A logarithm is basically solving for exponents, x in the following • 10x=1000 • here x = 3 • log101000 = x • log101000 = 3 • "The log of 1000, base 10, is 3”logbase(number) = exponent
So for pH… • If [H+] = 1.0 x 10-7 • and pH = - log [H+] • then pH = - log [1.0 x 10-7] = - (-7) log 10 • and pH = 7 x 1 = 7 • so this would be a neutral solution.
The pOH scale • pOH is the negative logarithm of the hydroxide ion concentration of a solution. pH = –log [OH-] • pH + pOH = 14
Concentration of Strong Acids & Bases For all strong monoprotic acids (ones that have one H+ ion to donate) the concentration of the acid is the concentration of H+ ions. For all strong bases, the concentration of the OH– ions is the concentration of the base. A 0.6M solution of NaOH means there are 0.6 moles of NaOH per liter of water. NaOH Na+ + OH- For every mole of NaOH, there is one mole of OH-, so the concentration is the same.
Neutralization • What happens if we mix a strong acid and a strong base? • A neutralization reactionis a reaction in which an acid and a base in an aqueous solution react to produce a salt and water. • The net reaction (without the salt) is:
Titration • It’s all in the technique…
Titration • Titrationis a method for determining the concentration of a solution by reacting a known volume of that solution with a solution of known concentration. • HCl + NaOH NaCl + H2O • If I have 50 mL of a 0.5M solution of HCl, how much of a 0.5M solution of NaOH should be needed to neutralize it? • How can we use this to determine the concentration of an unknown acid or base?
Titration Procedure • In a titration procedure, a measured volume of an acid or base of unknown concentration is placed in a beaker, and initial pH recorded.
Titration Procedure • A buret is filled with the titrating solution of known concentration, called a titrant.
Titration Procedure • Measured volumes of the titrant are added slowly and mixed into the solution in the beaker. • The pH is read and recorded after each addition.
pH indicator solution • Chemical dyes whose color are affected by acidic and basic solutions are called acid-base indicators. • An end point is the point at which an indicator used in a titration changes color. • An indicator will change color at the equivalence point.
End Point • The process continues until the reaction reaches the equivalence point, which is the point at which moles of H+ ions from the acid equals moles of OH– ions from the base. • An abrupt change in pH occurs at the equivalence point.
Naming Bases • Bases • Group 1 and 2 hydroxides… • NH3 (ammonia)
Naming Acids • Binary Acids (hydrogen + 1 element) • Prefix “Hydro” to name the hydrogen part of the compound • The rest of the word consists of a form of the root of the second element, plus the suffix “ic” • Second word is always “acid” • Example: HCl Hydrochloric Acid • Example: HF Hydrofluoric Acid • Example: HI • Example: HBr
Naming Acids • Oxyacids (contain hydrogen and oxyanion) • Identify oxyanion. The first word is the oxyanion name and a suffix • If original suffix was “ate” “ic” • If original suffix was “ite” “ous” • Second word = “acid” • Example: HClO4 = Perchloric Acid • Example: HClO3= Chloric Acid • Example: HClO2 = • Example: HClO= (adds chlorine to pools) • Example: HNO3 • Example: HNO2
In Conclusion • Using the pH scale, identify what makes a substances acidic, basic or neutral • What is more dangerous, an acid or a base? • What ion is responsible for an acidic solution? A basic solution? • Name the following acids • H2SO4 • H2SO3 • HCl • HF • H2CO3