Optical Ion Sensor
Optical Ion Sensor. Cheri W. Clavier , Yihui Yang, Alicia Vogt, D. Lynn Rodman, Joseph F. Sinski, Hee-Jung Im, and Ziling Xue* Department of Chemistry, University of Tennessee Knoxville, TN 37996. Abstract.

Optical Ion Sensor
E N D
Presentation Transcript
Optical Ion Sensor Cheri W. Clavier, Yihui Yang, Alicia Vogt, D. Lynn Rodman, Joseph F. Sinski, Hee-Jung Im, and Ziling Xue* Department of Chemistry, University of Tennessee Knoxville, TN 37996
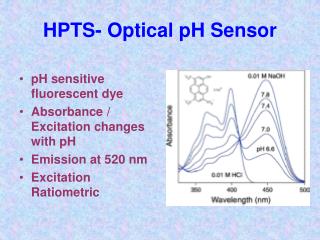
Abstract Heavy metal ions are used in a variety of industrial applications and many are considered pollutants. Currently, industrial metal ion concentration measurements are mostly determined off-line, using techniques that are usually costly, time-consuming and inconvenient. We are developing an optical ion sensor through the sol-gel process that will give concentrations of metal ions. Initial work has been carried out to explore the basic properties of sol-gel glasses with various amounts of amine ligand incorporated directly into the sol-gel matrix. The sol-gel preparation process was modified to produce optically transparent monoliths containing either the ethylenediamine derivative NH2(CH2)2NH(CH2)3Si(OMe3)3 (TMSen) or the monoamine ligand HN2(CH2)3Si(OMe)3 using Si(OMe)4 as a cross-linking agent. We have developed a novel gel-preparation process that gives strong, optically transparent monoliths without cracking. In addition, a sensor holder has been designed in a flow cell system that allows spectrophotometric measurements of the sol-gel disk in flowing Cu2+ solutions. The kinetics of cupric ion uptake by the gels in solutions of various Cu2+ concentrations was studied by measuring the change in the absorbance of the metal:ligand complex over time using UV-VIS spectrophotometry. The gels are regenerated with acid or EDTA, neutralized, and then used in subsequent Cu2+ uptake/ removal cycles. Our studies thus far show the potential of the system in metal ion sensing applications. We are currently focused on improving the gel regeneration procedure to provide reproducible second, third, and fourth cycle results.
Motivation for Project • Cu2+ is used in a variety of industrial applications such as microelectronics, piping, and electroplating; • Cu2+ is recognized by the EPA as a toxic metal pollutant; • Concentrations of Cu2+ in industrial waste can vary widely, ranging from 0.5-500 ppm; • Currently, Cu2+ concentration measurements are determined off-line.
Purpose - Long Term Goals • Chemically graft ligands onto sol-gel matrices and prepare transparent monoliths suitable for optical analysis; • Evaluate the spectroscopic response of such ligand-grafted sol-gels to target metal ion concentrations; • Study the stability and durability of the sol-gel sensors; • Test in flow systems; • Investigate the influence of other chemicals.
Related Studies • Sol-gel probes (single-use only) have been reported for the determination of other heavy metal ions;1 • Diamine (TMSen) anchored sol-gels have shown selective intake of Cu2+ ion with favorable kinetics and easy removal;2 • Optically transparent, stable sol-gel monoliths have been reported using bis-TMSen anchored sol-gels for the analysis of proteins.3 • (1) Oheme, I.; Wolfbeis, O.S. Mikrochim. Acta 1997, 126, 177-192 • (2) Im, H.-J.; Yang, Y.; Allain, L. R.; Barnes, C. E.; Dai, S.; Xue, Z. Environ. Sci. Technol. 2000, 34, 2209-2214 • (3) Rao, M. S.; Dave, B. C. J. Am. Chem. Soc. 1998, 120, 13270-13271
Our Approaches • Select ligands with good binding affinities for target metal ions, such as ethylenediamine NH2(CH2)2NH-(CH2)3Si(OMe)3 or monoamine NH2(CH2)3Si(OMe)3 derivatives for Cu2+; • Incorporate the ligands into a sol-gel using Si(OR)4 (TMOS) as a cross-linking agent.
Hydrolysis Condensation Functionalized ligand, L-Si(OR)3, can be anchored into the sol-gel matrix; The ligand, L-, is covalently bound to the gel; Ligand leaching is not a problem. The Sol-Gel Process
Current Method Form gel disks free of amine ligand; Graft ligand in the 2nd step; Use ethylene glycol to enhance gel strength; Develop base-catalyzed process. Advantages Fewer cracks; Stronger gel disks for flow cell operation and Cu2+ uptake-removal cycles; Optically transparent sensors. Novel Gel Preparation
Characterization Studies Brunauer-Emmett-Teller (BET) Gas Adsorption • Narrow pore size distribution; • Average pore diameter: • Blank Gels: 45 Å • Ligand-Grafted Gels: 67 Å • Fast kinetics.
Flow-Cell Design • Accommodate and hold gel disks of various thickness steady in flowing solution; • Support two fiber optic cables that are isolated from the solution; • Interface with Ocean Optics S2000 fiber optic spectrometer.
Regeneration:Diamine-grafted gels • Procedure: • Cu2+ is easily removed from the gels with 1.0 M HCl; • 0.010 M NaOH may be used to neutralize the gel. • Gel regeneration could be done on-line using the flow cell; • Abs vs. time plots change from first to second cycle.
Reproducibility Study:Diamine-grafted gels • 0.0777 ± 0.012 • One cycle • 50.01 mM [Cu2+]
Results:Diamine-grafted gels • Showed a correlation between slope dA/dt and [Cu2+] for the 1st cycle; • Could be regenerated with acid; • Neutralization with base was difficult; • Results not reproducible from the 1st to 2nd cycle; • Potential use as a disposable Cu2+ probe.
Reproducibility Study:Monoamine-grafted gels • Five cycles • 75.03 mM [Cu2+]
Diamine gel Two-step preparation; Not reproducible from the 1st to 2nd cycle; Metal ion probe - one time use. Monoamine gel Four-step imprinting preparation; Reproducible over at least 5 cycles; Metal ion sensor for multi-cycle use. Comparison - Diamine vs. Monoamine Gels
Results • Prepared transparent sol-gel monoliths as substrates; • Successfully grafted amine ligands into the sol-gel matrix; • Designed a flow-cell coupled to a portable spectrophotometer; • Evaluated the spectroscopic response of ligand-grafted sol-gels to target metal ion concentrations; • Demonstrated the application of diamine ligand-grafted gels as a disposable Cu2+ probe; • Illustrated the use of monoamine ligand-grafted gels as Cu2+ sensors for multi-cycle use.
Future Plans • Investigate the use of new, more sensitive ligands for different metal ions; • Explore thin film sensor approach for faster response; • Study the influence of other chemicals; • Explore “cocktail” approach for multi-component analysis; • Conduct field tests; • Explore on-line regeneration of sensors.
Acknowledgements • Measurement and Control Engineering Center • University of Tennessee, Knoxville