Soil water
580 likes | 597 Views
Learn about soil water terms like infiltration, percolation, evapotranspiration, and more. Discover the types of water in soil and their availability to plants. Understand critical levels of water in soil and the forces influencing soil water movement.

Soil water
E N D
Presentation Transcript
Some terms • Infiltration • Rate depends on texture and topography • Percolation • Water table • Evapotranspiration • Runoff
Water forms films on soil particles Water is polar; soil particle surfaces are negatively charged.
Types of water in soil: • 1. Adhesion water • “HYGROSCOPIC WATER” • Remove by oven drying • Not available to plants
Hygroscopic coefficient(an amount of water) • Amount of moisture in air dry soil • Difference between air dry and oven dry amounts
2. Cohesion water • “CAPILLARY WATER” • 15 – 20 molecules thick • Remove by air drying • Most is available to plants • some unavailable to plants (especially in clay or high OM soils)
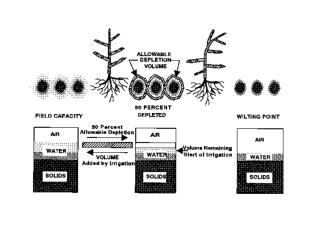
WILTING POINT(an amount of water) “the amount of water in the soil when plants have removed all that they can” Divides available and unavailable water
Wilting Point • Amount of water in soil when plants begin to wilt.
Difference between wilting point and hygroscopic coefficient: at hygroscopic coefficient at wilting point Moist Dry to touch Can’t squeeze water Air-dried Plant can’t get water Can be oven dried to remove water
3. Gravitational water • Not available to plants • Drains through soil under influence of gravity • Through large pores • Small pores can hold water against pull of gravity through capillarity
FIELD CAPACITY(an amount of water) “amount of water after gravity has removed all freely drained water” Divides capillary water from gravitational water
Field Capacity • Amount of water in soil after free drainage has removed gravitational water (2 – 3 days) • Soil is holding maximum amount of water available to plants • Optimal aeration (micropores filled with water; macropores with air)
(Air dry) Field capacity Oven dry Hydroscopic coefficient Wilting point No water Plant-unavailable water (capillary) Plant-available Water (capillary) Adhesion water Gravitational water
capillarity Height water will rise in cylinder depends on diameter of tube; due to adhesion of water and tube Plastic Glass
Critical levels of water in soil: • Field capacity • Wilting point • Hygroscopic coefficient Plant available water is between field capacity and wilting point.
Not all capillary water is equally available to plants • Plants can extract water easily from soils that are near field capacity • Sponge example • Wilting point is not the same for all plants • Sunflowers can extract more water from soil than corn • Sponge example
Micropores full; macropores have air Adhesion water Wilting point Field Capacity All pores full Gravitational water
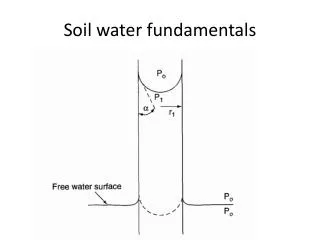
Hydraulic pressure of soil water • Pressure = force / area Hydraulic pressure “0” at surface increases with depth Open body of water
Same in saturated soil “0” at surface increases with depth
Capillary pressure • Thin tube in open pan water (Adhesion to walls of tube; cohesion in center of tube; therefore thin tube only) -20 g/cm3 Pressure in tube decreases away from water surface -10 0
Same in unsaturated soil: • Capillary water is water in small pores continuously connected to free water surface (soil water table) -20 Capillary water (continuous film) -10 0 Soil water table Saturated soil +10
the smaller the pore space, the higher capillary water will rise in profile • Smaller pore space, tighter water is held to particle surfaces against gravity (i.e., higher field capacity) clay silt sand Pan of water
at • Insert Fig 9.6
Energy status of soil water • Energy status • Things move to lower energy states • It takes work to keep them from doing so • E.g. keeping something from falling in response to gravity • Influences water movement • E.g. adhesion attracts water to soil particles so particles close to soil are at lower energy state
Forces on soil water: • Adhesion • Attracts water to soil particles • Holds adhesion(hygroscopic) water and cohesion (capillary) water • Called “matric force” • Ions in solution • Attracts water to ions • Called “osmotic force” • Gravity • Pulls water downward • “gravitational force”
Soil water potential • Amount of work required to move water • Expressed in bars or Pascals • Similar to soil water tension
potentials Water is removed by various potentials
Water moves from areas of higher water potential (wetter) to areas of lower water potential (drier).
Potentials • Matric • Gravitational • Hydrostatic • Osmotic
Matric potential • Work required to remove water held by adhesion to soil surface and cohesion in capillary pores. • Hygroscopic and capillary water
Gravitational potential • Work required to draw water down in response to gravity • Applies to gravitational water only
Hydrostatic Potential Work required to move water below the water table; applies only to saturated conditions
Osmotic potential If there are solutes in the solution, water will group around them and reduce the freedom of water movement, i.e., lowering the potential.
Osmotic potential • Water containing salts is less able to do work than pure water • e.g., cannot boil at standard boiling point • The more salts, the lower the potential • Important for plant uptake • In “salty” soil, potential in soil solution may be lower than inside plant root cells, impeding ability of water to pass into plant
Practical application • Irrigation water contains soluble salts. • When water evapotranpirates, salts stay behind in soil. • They can be removed by adequate rainfall that flushes the salts to the water table. • BUT • If water table is too high, salts cannot be flushed. • If climate is arid, salts cannot be flushed by rainfall. • If soil is fine-textured or has poor structure, water will not be flushed. • Creates SALINE soils
SALINE soils • Too much salt in soil water prevents plants from getting water. • Salinity raises osmotic pressure of soil water inhibiting water uptake. • Applying too much fertilizer has same effect because fertilizers present nutrients in the form of “salts”