Chapter 25 Metabolism
Chapter 25 Metabolism. Functions of food source of energy essential nutrients stored for future use Metabolism is all the chemical reactions of the body some reactions produce the energy stored in ATP that other reactions consume

Chapter 25 Metabolism
E N D
Presentation Transcript
Chapter 25Metabolism • Functions of food • source of energy • essential nutrients • stored for future use • Metabolism is all the chemical reactions of the body • some reactions produce the energy stored in ATP that other reactions consume • all molecules will eventually be broken down and recycled or excreted from the body Tortora & Grabowski 9/e 2000 JWS
Catabolism and Anabolism • Catabolic reactions breakdown complex organic compounds • providing energy (exergonic) • glycolysis, Krebs cycle and electron transport • Anabolic reactions synthesize complex molecules from small molecules • requiring energy (endergonic) • Exchange of energy requires use of ATP (adenosine triphosphate) molecule. Tortora & Grabowski 9/e 2000 JWS
ATP Molecule & Energy • Each cell has about 1 billion ATP molecules that last for less than one minute • Over half of the energy released from ATP is converted to heat Tortora & Grabowski 9/e 2000 JWS
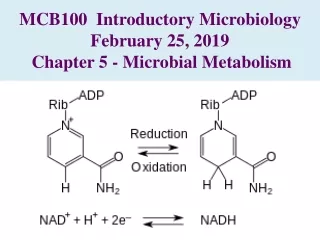
Energy Transfer • Energy is found in the bondsbetween atoms • Oxidation is a decrease in the energy content of a molecule • Reduction is the increase in the energy content of a molecule • Oxidation-reduction reactions are always coupled within the body • whenever a substance is oxidized, another is almost simultaneously reduced. Tortora & Grabowski 9/e 2000 JWS
Oxidation and Reduction • Biological oxidation involves the loss of (electrons) hydrogen atoms • dehydrogenation reactions require coenzymes to transfer hydrogen atoms to another compound • common coenzymes of living cells that carry H+ • NAD (nicotinamide adenine dinucleotide ) • NADP (nicotinamide adenine dinucleotide phosphate ) • FAD (flavin adenine dinucleotide ) • Biological reduction is the addition of electrons (hydrogen atoms) to a molecule • increase in potential energy of the molecule Tortora & Grabowski 9/e 2000 JWS
Mechanisms of ATP Generation ADP + P = ATP • Phosphorylation is • bond attaching 3rd phosphate group contains stored energy • Mechanisms of phosphorylation • within animals • substrate-level phosphorylation in cytosol • oxidative phosphorylation in mitochondria • in chlorophyll-containing plants or bacteria • photophosphorylation. Tortora & Grabowski 9/e 2000 JWS
Phosphorylation in Animal Cells • In cytoplasm (1) • In mitochondria (2, 3 & 4) Tortora & Grabowski 9/e 2000 JWS
Carbohydrate Metabolism--In Review • In GI tract • polysaccharides broken down into simple sugars • absorption of simple sugars (glucose, fructose & galactose) • In liver • fructose & galactose transformed into glucose • storage of glycogen (also in muscle) • In body cells --functions of glucose • oxidized to produce energy • conversion into something else • storage energy as triglyceride in fat Tortora & Grabowski 9/e 2000 JWS
Fate of Glucose • ATP production during cell respiration • uses glucose preferentially • Converted to one of several amino acids in many different cells throughout the body • Glycogenesis • hundreds of glucose molecules combined to form glycogen for storage in liver & skeletal muscles • Lipogenesis (triglyceride synthesis) • converted to glycerol & fatty acids within liver & sent to fat cells Tortora & Grabowski 9/e 2000 JWS
Glucose Movement into Cells • In GI tract and kidney tubules, Na+/glucose symporters • Most other cells, GluT facilitated diffusion transporters move glucose into cells • insulin increases number of GluT transporters in the membrane of most cells • in liver & brain, always lots of GluT transporters • Glucose 6-phosphate forms immediately inside cell (requires ATP) thus, glucose hidden in cell • Concentration gradient favorable for more glucose to enter Tortora & Grabowski 9/e 2000 JWS
Glucose Catabolism • Cellular respiration • 4 steps are involved • glucose + O2 producesH2O + energy + CO2 • Anaerobic respiration • called glycolysis (1) • formation of acetyl CoA (2)is transitional step to Krebs cycle • Aerobic respiration • Krebs cycle (3) and electron transport chain (4) Tortora & Grabowski 9/e 2000 JWS
Glycolysis of Glucose & Fate of Pyruvic Acid • Breakdown of six-carbon glucose molecule into 2 three-carbon molecules of pyruvic acid • 10 step process occurring in cell cytosol • produces 4 molecules of ATP after input of 2 ATP • utilizes 2 NAD+ molecules as hydrogen acceptors • If O2 shortage in a cell • pyruvic acid is reduced to lactic acid so that NAD+ will be still available for further glycolysis • rapidly diffuses out of cell to blood • liver cells remove it from blood & convert it back to pyruvic acid Tortora & Grabowski 9/e 2000 JWS
10 Steps of Glycolysis Tortora & Grabowski 9/e 2000 JWS
Formation of Acetyl Coenzyme A • Pyruvic acid enters the mitochondria with help of transporter protein • Decarboxylation • pyruvate dehydrogenase converts 3 carbon pyruvic acid to 2 carbon fragment (CO2 produced) • pyruvic acid was oxidized so that NAD+ becomes NADH • 2 carbon fragment (acetyl group) is attached to Coenzyme A to form Acetyl coenzyme A which enter Krebs cycle • coenzyme A is derived from pantothenic acid (B vitamin). Tortora & Grabowski 9/e 2000 JWS
Krebs Cycle (Citric Acid Cycle) • Series of oxidation-reduction & decarboxylation reactions occurring in matrix of mitochondria • It finishes the same as it starts (4C) • acetyl CoA (2C) enters at top & combines with a 4C compound • 2 decarboxylation reactions peel 2 carbons off again when CO2 is formed Tortora & Grabowski 9/e 2000 JWS
Krebs Cycle • Energy stored in bonds is released step by step to form several reduced coenzymes (NADH & FADH2) that store the energy • In summary: each Acetyl CoAmolecule that enters the Krebscycle produces • 2 molecules of C02 • one reason O2 is needed • 3 molecules of NADH + H+ • one molecule of ATP • one molecule of FADH2 • Remember, each glucoseproduced 2 acetyl CoA molecules Tortora & Grabowski 9/e 2000 JWS
The Electron Transport Chain • Series of integral membrane proteins in the inner mitochondrial membrane capable of oxidation/reduction • Each electron carrier is reduced as it picks up electrons and is oxidized as it gives up electrons • Small amounts of energy released in small steps • Energy used to form ATP by chemiosmosis Tortora & Grabowski 9/e 2000 JWS
Chemiosmosis • Small amounts of energy released as substances are passed along inner membrane • Energy used to pump H+ ions from matrix into space between inner & outer membrane • High concentration of H+ is maintained outside of inner membrane • ATP synthesis occurs as H+ diffuses through a special H+ channel in inner membrane Tortora & Grabowski 9/e 2000 JWS
Electron Carriers • Flavin mononucleotide (FMN) is derived from riboflavin (vitamin B2) • Cytochromes are proteins with heme group (iron) existing either in reduced form (Fe+2) or oxidized form (Fe+3) • Iron-sulfur centers contain 2 or 4 iron atoms bound to sulfur within a protein • Copper (Cu) atoms bound to protein • Coenzyme Q is nonprotein carrier mobile in the lipid bilayer of the inner membrane Tortora & Grabowski 9/e 2000 JWS
Steps in Electron Transport • Carriers of electron transport chain are clustered into 3 complexes that each act as proton pump (expel H+) • Mobile shuttles pass electrons between complexes • Last complex passes its electrons (2H+) to a half of O2 molecule to form a water molecule (H2O) Tortora & Grabowski 9/e 2000 JWS
Proton Motive Force & Chemiosmosis • Buildup of H+ outside the inner membrane creates + charge • electrochemical gradient potential energy is called proton motive force • ATP synthase enzyme within H+ channel uses proton motive force to synthesize ATP from ADP and P Tortora & Grabowski 9/e 2000 JWS
Summary of Cellular Respiration • Glucose + O2 is broken down into CO2 + H2O + energy used to form 36 to 38 ATPs • 2 ATP are formed during glycolysis • 2 ATP are formed by phosphorylation during Krebs cycle • electron transfers in transport chain generate 32 or 34 ATPs from one glucose molecule • Summary in Table 25.1 • Points to remember • ATP must be transported out of mitochondria in exchange for ADP • uses up some of proton motive force • Oxygen is required or many of these steps can not occur Tortora & Grabowski 9/e 2000 JWS
Carbohydrate Loading • Long-term athletic events (marathons) can exhaust glycogen stored in liver and skeletal muscles • Eating large amounts of complex carbohydrates (pasta & potatoes) for 3 days before a marathon maximizes glycogen available for ATP production • Useful for athletic events lasting for more than an hour Tortora & Grabowski 9/e 2000 JWS
Glycogenesis & Glycogenolysis • Glycogenesis • glucose storage as glycogen • 4 steps to glycogenformation in liver orskeletal muscle • stimulated by insulin • Glycogenolysis • glucose release not a simplereversal of steps • enzyme phosphorylase splits off a glucose molecule by phosphorylation to form glucose 1-phosphate • enzyme only in hepatocytes so muscle can’t release glucose • enzyme activated by glucagon (pancreas) & epinephrine (adrenal) Tortora & Grabowski 9/e 2000 JWS
Gluconeogenesis • Liver glycogen runs low if fasting, starving or not eating carbohydrates forcing formation from other substances • lactic acid, glycerol & certain amino acids (60% of available) • Stimulated by cortisol (adrenal) & glucagon (pancreas) • cortisol stimulates breakdown of proteins freeing amino acids • thyroid mobilizes triglycerides from adipose tissue Tortora & Grabowski 9/e 2000 JWS
Transport of Lipids by Lipoproteins • Most lipids are nonpolar and must be combined with protein to be tranported in blood • Lipoproteins are spheres containing hundreds of molecules • outer shell polar proteins(apoproteins) & phospholipids • inner core of triglyceride & cholesterol esters • Lipoprotein categorized byfunction & density • 4 major classes of lipoproteins • chylomicrons, very low-density, low-density & high-density lipoproteins Tortora & Grabowski 9/e 2000 JWS
Classes of Lipoproteins • Chylomicrons (2 % protein) • form in intestinal epithelial cells to transport dietary fat • apo C-2 activates enzyme that releases the fatty acids from the chylomicron for absorption by adipose & muscle cells • liver processes what is left • VLDLs (10% protein) • transport triglycerides formed in liver to fat cells • LDLs (25% protein) --- “bad cholesterol” • carry 75% of blood cholesterol to body cells • apo B100 is docking protein for receptor-mediated endocytosis of the LDL into a body cell • if cells have insufficient receptors, remains in blood and more likely to deposit cholesterol in artery walls (plaque) • HDLs (40% protein) --- “good cholesterol” • carry cholesterol from cells to liver for elimination Tortora & Grabowski 9/e 2000 JWS
Blood Cholesterol • Sources of cholesterol in the body • food (eggs, dairy, organ meats, meat) • synthesized by the liver • All fatty foods still raise blood cholesterol • liver uses them to create cholesterol • stimulate reuptake of cholesterol containing bile normally lost in the feces • Desirable readings for adults • total cholesterol under 200 mg/dL; triglycerides 10-190 mg/dL • LDL under 130 mg/dL; HDL over 40 mg/dL • cholesterol/HDL ratio above 4 is undesirable risk • Raising HDL & lowering cholesterol can be accomplished by exercise, diet & drugs Tortora & Grabowski 9/e 2000 JWS
Fate of Lipids • Oxidized to produce ATP • Excess stored in adipose tissue or liver • Synthesize structural or important molecules • phospholipids of plasma membranes • lipoproteins that transport cholesterol • thromboplastin for blood clotting • myelin sheaths to speed up nerve conduction • cholesterol used to synthesize bile salts and steroid hormones. Tortora & Grabowski 9/e 2000 JWS
Triglyceride Storage • Adipose tissue removes triglycerides from chylomicrons and VLDL and stores it • 50% subcutaneous, 12% near kidneys, 15% in omenta, 15% in genital area, 8% between muscles • Fats in adipose tissue are ever-changing • released, transported & deposited in other adipose • Triglycerides store more easily than glycogen • do not exert osmotic pressure on cell membranes • are hydrophobic Tortora & Grabowski 9/e 2000 JWS
Lipid Catabolism: Lipolysis & Glycerol • Triglycerides are split into fatty acids & glycerol by lipase • glycerol • if cell ATP levels are high, converted into glucose • if cell ATP levels are low, converted into pyruvic acid which enters aerobic pathway to ATP production Tortora & Grabowski 9/e 2000 JWS
Lipolysis & Fatty acids Liver cells • Beta oxidation in mitochondria removes 2 carbon units from fatty acid & forms acetyl coenzyme A • Liver cells form acetoacetic acid from 2 carbon units & ketone bodies from acetoacetic acid (ketogenesis) • heart muscle & kidney cortex prefer to use acetoacetic acid for ATP production Tortora & Grabowski 9/e 2000 JWS
Lipid Anabolism: Lipogenesis • Synthesis of lipids by liver cells = lipogenesis • from amino acids • converted to acetyl CoA & then to triglycerides • from glucose • from glyceraldehyde 3-phosphate to triglycerides • Stimulated by insulin when eat excess calories Tortora & Grabowski 9/e 2000 JWS
Ketosis • Blood ketone levels are usually very low • many tissues use ketone for ATP production • Fasting, starving or high fat meal with few carbohydrates results in excessive beta oxidation & ketone production • acidosis (ketoacidosis) is abnormally low blood pH • sweet smell of ketone body acetone on breath • occurs in diabetic since triglycerides are used for ATP production instead of glucose & insulin inhibits lipolysis Tortora & Grabowski 9/e 2000 JWS
Fate of Proteins • Proteins are broken down into amino acids • transported to the liver • Usage • oxidized to produce ATP • used to synthesize new proteins • enzymes, hemoglobin, antibodies, hormones, fibrinogen, actin, myosin, collagen, elastin & keratin • excess converted into glucose or triglycerides • no storage is possible • Absorption into body cells is stimulated by insulinlike growth factors (IGFs) & insulin Tortora & Grabowski 9/e 2000 JWS
Protein Catabolism • Breakdown of protein into amino acids • Liver cells convert amino acids into substances that can enter the Krebs cycle • deamination removes the amino group (NH2) • converts it to ammonia (NH3) & then urea • urea excreted in the urine • Converted substances enter the Krebs cycle to produce ATP Tortora & Grabowski 9/e 2000 JWS
Protein Anabolism • Production of new proteins by formation of peptide bonds between amino acids • 10 essential amino acids are ones we must eat because we can not synthesize them • nonessential amino acids can be synthesized by transamination (transfer of an amino group to a substance to create an amino acid) • Occurs on ribosomes in almost every cell • Stimulated by insulinlike growth factor, thyroid hormone, insulin, estrogen & testosterone • Large amounts of protein in the diet do not cause the growth of muscle, only weight-bearing exercise Tortora & Grabowski 9/e 2000 JWS
Phenylketonuria (PKU) • Genetic error of protein metabolism that produces elevated blood levels of amino acid phenylalanine • causes vomiting, seizures & mental retardation • normally converted by an enzyme into tyrosine which can enter the krebs cycle • Screening of newborns prevents retardation • spend their life with a diet restricting phenylalanine • restrict Nutrasweet which contains phenylalanine Tortora & Grabowski 9/e 2000 JWS
Key Molecules at Metabolic Crossroads • Glucose 6-phosphate, pyruvic acid and acetyl coenzyme A play pivotal roles in metabolism • Different reactions occur because of nutritional status or level of physical activity Tortora & Grabowski 9/e 2000 JWS
Role of Glucose 6-Phosphate • Glucose is converted to glucose 6-phosphate just after entering the cell • Possible fates of glucose 6-phosphate • used to synthesize glycogen when glucose is abundant • if glucose 6-phosphatase is present, glucose can be re-released from the cell • precursor of a five-carbon sugar used to make RNA & DNA • converted to pyruvic acid during glycolysis in most cells of the body Tortora & Grabowski 9/e 2000 JWS
Role of Pyruvic Acid • 3-carbon molecule formed when glucose undergoes glycolysis • If oxygen is available, cellular respiration proceeds • If oxygen is not available, only anaerobic reactions can occur • pyruvic acid is changed to lactic acid • Conversions • amino acid alanine produced from pyruvic acid • to oxaloacetic acid of Krebs cycle Tortora & Grabowski 9/e 2000 JWS
Role of Acetyl coenzyme A • Can be used to synthesizefatty acids, ketone bodies, or cholesterol • Can not be converted to pyruvic acid so can not be used to reform glucose Tortora & Grabowski 9/e 2000 JWS
Metabolic Adaptations • Absorptive state • nutrients entering the bloodstream • glucose readily available for ATP production • 4 hours for absorption of each meal so absorptive state lasts for 12 hours/day • Postabsorptive state • absorption of nutrients from GI tract is complete • body must meet its needs without outside nutrients • late morning, late afternoon & most of the evening • assuming no snacks, lasts about 12 hours/day • more cells use ketone bodies for ATP production • maintaining a steady blood glucose level is critical Tortora & Grabowski 9/e 2000 JWS
Metabolism during Absorptive State • Body cells use glucose for ATP production • about 50% of absorbed glucose • Storage of excess fuels occur in hepatocytes, adipocytes & skeletal muscle • most glucose entering liver cells is converted to glycogen (10%) or triglycerides (40%) • dietary lipids are stored in adipose tissue • amino acids are deaminated to enter Krebs cycle or are converted to glucose or fatty acids • amino acids not taken up by hepatocytes used by other cells for synthesis of proteins Tortora & Grabowski 9/e 2000 JWS
Absorptive State Points where insulin stimulation occurs. Tortora & Grabowski 9/e 2000 JWS
Regulation of Metabolism during Absorptive State • Beta cells of pancreas release insulin • Insulin’s functions • increases anabolism & synthesis of storage molecules • decreases catabolic or breakdown reactions • promotes entry of glucose & amino acids into cells • stimulates phosphorylation of glucose • enhances synthesis of triglycerides • stimulates protein synthesis along with thyroid & growth hormone Tortora & Grabowski 9/e 2000 JWS
Metabolism During Postabsorptive State • Maintaining normal blood glucose level (70 to 110 mg/100 ml of blood) is major challenge • glucose enters blood from 3 major sources • glycogen breakdown in liver produces glucose • glycerol from adipose converted by liver into glucose • gluconeogenesis using amino acids produces glucose • alternative fuel sources are • fatty acids from fat tissue fed into Krebs as acetyl CoA • lactic acid produced anaerobically during exercise • oxidation of ketone bodies by heart & kidney • Most body tissue switch to utilizing fatty acids, except brain still need glucose. Tortora & Grabowski 9/e 2000 JWS
Postabsorptive State Tortora & Grabowski 9/e 2000 JWS
Regulation of Metabolism During Postabsorptive State • As blood glucose level declines, pancreatic alpha cells release glucagon • glucagon stimulates gluconeogenesis & glycogenolysis within the liver • Hypothalamus detects low blood sugar • sympathetic neurons release norepinephrine and adrenal medulla releases norepinephrine & epinephrine • stimulates glycogen breakdown & lipolysis • raises glucose & free fatty acid blood levels Tortora & Grabowski 9/e 2000 JWS
Metabolism During Fasting & Starvation • Fasting means going without food for hours/days • Starvation means weeks or months • can survive 2 months or more if drink enough water • amount of adipose tissue is determining factor • Nutritional needs • nervous tissue & RBC need glucose so amino acids will be broken down for gluconeogenesis • blood glucose stabilizes at 65 mg/100 mL • lipolysis releases glycerol used in gluconeogenesis • increase in formation of ketone bodies by liver cells due to catabolism of fatty acids • by 40 days, ketones supply 2/3’s of brains fuel for ATP Tortora & Grabowski 9/e 2000 JWS