Topic 5: Gases
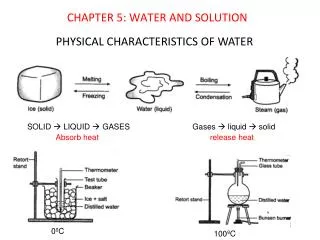
Topic 5: Gases. Pressure. Force per unit area. Gas molecules fill container. Molecules move around and hit sides. The force is caused by collisions with the sides of the container. Container has the area. Measured with a barometer. Barometer. Vacuum.

Topic 5: Gases
E N D
Presentation Transcript
Pressure • Force per unit area. • Gas molecules fill container. • Molecules move around and hit sides. • The force is caused by collisions with the sides of the container. • Container has the area. • Measured with a barometer.
Barometer Vacuum • The pressure of the atmosphere at sea level will hold a column of mercury 760 mm Hg. • 1 atm = 760 mm Hg 760 mm Hg 1 atm Pressure
Manometer • The column of mercury indicates the pressure. • “h” (height in mm) indicates how much lower the pressure in the container is compared to the atmosphere. h Gas
Manometer • “h” (height in mm) indicates how much higher the pressure in the container is compared to the atmosphere. h Gas
Units of pressure • 1 atmosphere = 760 mm Hg • 1 mm Hg = 1 torr • 1 atm = 101,235 Pascals = 101.325 kPa • Can make conversion factors from these. • What is 724 mm Hg in kPa? • in torr? • in atm?
The Gas Laws • Boyle’s Law • Pressure and volume are inversely related at constant temperature. • PV= k • As one goes up, the other goes down. • P1V1 = P2 V2 • Graphically
V P (at constant T)
Slope = k V 1/P (at constant T)
22.41 L atm O2 PV CO2 P (at constant T)
Examples • Sulfur dioxide (SO2), a gas that plays a central role in the formation of acid rain, is found in the exhaust of automobiles and power plants. • Consider a 1.53-L sample of gaseous SO2 at a pressure of 5.6 x 103 Pa. If the pressure is changed to 1.5 x 104 Pa at a constant temperature, what will be the new volume?
Charle’s Law • Volume of a gas varies directly with the absolute temperature at constant pressure. • V = kT (if T is in Kelvin) • V1 = V2 T1 = T2 • Graphically it appears as follows:
He CH4 H2O V (L) H2 -273.15ºC T (ºC)
Examples • A sample of gas at 15oC and 1 atm has a volume of 2.58 L. What volume will this gas occupy at 38oC and 1 atm?
Avogadro's Law • At constant temperature and pressure, the volume of gas is directly related to the number of moles. • V = k n (n is the number of moles) • V1 = V2 • n1n2
Suppose we have a 12.2-L sample containing 0.50 mol of oxygen gas (O2) at a pressure of 1 atm and a temperature of 25oC. • If all this O2 were converted to ozone (O3) at the same temperature and pressure, what would be the volume of the ozone?
Ideal Gas Law • PV = nRT • V = 22.41 L at 1 atm, 0ºC, n = 1 mole, what is R? • R is the ideal gas constant. • R = 0.08206 L ∙ atm / K ∙ mol • Tells you about a gas at a given time. • The other laws tell you about a gas when it changes.
Ideal Gas Law • An equation of state. • Independent of how it gets to that state. Does not depend on the path. • Given 3 variables you can determine the fourth. • An Empirical Equation - based on experimental evidence.
Ideal Gas Law • A hypothetical substance - the ideal gas • Gases only approach ideal behavior at low pressure (< 1 atm) and high temperature. • Use the Ideal gas law anyway, unless told to do otherwise. • It gives good estimates.
Examples • A sample of hydrogen gas (H2) has a volume of 8.56 L at a temperature of 0oC and a pressure of 1.5 atm. Calculate the moles of H2 molecules present in this gas sample. • Suppose we have a sample of ammonia gas with a volume of 7.0 mL at a pressure of 1.68 atm. The gas is compressed to a volume of 2.7 mL at a constant temperature. Use the ideal gas law to calculate the final pressure.
A sample of methane gas that has a volume of 3.8 L at 5oC is heated to 86oC at constant pressure. Calculate its new volume. • A sample of diborane gas (B2H6), a substance that bursts into flame when exposed to air, has a pressure of 345 torr at a temperature of -15oC and a volume of 3.48 L. If conditions are changed so that the temperature is 36oC and the pressure is 468 torr, what will be the volume of the sample?
Gases and Stoichiometry • Reactions happen in moles • At Standard Temperature and Pressure (STP, 0ºC and 1 atm) 1 mole of gas occupies 22.42 L. • If not at STP, use the ideal gas law to calculate moles of reactant or volume of product.
Examples • A sample of nitrogen gas has a volume of 1.75 L at STP. How many moles of N2 are present? • Quicklime (CaO) is produced by the thermal decomposition of calcium carbonate (CaCO3). Calculate the volume of CO2 at STP produced from the decomposition of 152g CaCO3 by the reaction • CaCO3 (s) → CaO (s) + CO2 (g)
Examples • A sample of methane gas having a volume of 2.80 L at 25oC and 1.65 atm was mixed with a sample of oxygen gas having a volume of 35.0 L at 31oC and 1.25 atm. The mixture was then ignited to form carbon dioxide and water. Calculate the volume of CO2 formed at a pressure of 2.50 atm and a temperature of 125oC.
Gas Density and Molar Mass • n = grams of gas = m molar mass molar mass • P = nRT = (m/molar mass) RT = m(RT) V VV(molar mass) Since d = m/V then P = dRT molar mass And … molar mass = dRT P
Example • The density of a gas was measured at 1.50 atm and 27oC and found to be 1.95 g/L. Calculate the molar mass of the gas.
Dalton’s Law • The total pressure in a container is the sum of the pressure each gas would exert if it were alone in the container. • The total pressure is the sum of the partial pressures. • PTotal = P1 + P2 + P3 + P4 + P5 ... • For each P = nRT V
Dalton's Law • PTotal = n1RT + n2RT + n3RT +... V VV • In the same container R, T and V are the same. • PTotal = (n1+ n2 + n3+...) RT V • PTotal = (nTotal) RT V
Example • Mixtures of helium and oxygen can be used in scuba diving tanks to help prevent “the bends.” For a particular dive, 46 L He at 25oC and 1.0 atm and 12 L O2 at 25oC and 1.0 atm were pumped into a tank with a volume of 5.0 L. Calculate the partial pressure of each gas and the total pressure in the tank at 25oC.
The mole fraction • Ratio of moles of the substance to the total moles. • symbol is Greek letter chi c • c1 = n1 = P1 • nTotalPtotal • P1 = c1 x PTotal
Examples • The partial pressure of oxygen was observed to be 156 torr in air with a total atomospheric pressure of 743 torr. Calculate the mole fraction of O2 present. • The mole fraction of nitrogen in the air is 0.7808. Calculate the partial pressure of N2 in air when the atmospheric pressure is 760 torr.
Examples • When these valves are opened, what is each partial pressure and the total pressure? 4.00 L CH4 1.50 L N2 3.50 L O2 0.752 atm 2.70 atm 4.58 atm
Vapor Pressure • When water evaporates, the vapor has a pressure. • Gases that are collected over water will have water vapor in them, so the vapor pressure of water must be subtracted from the total pressure. ( http://www.youtube.com/watch?v=ddw4fv391UY ) • The vapor pressure of water is either given or looked up.
Example • A sample of solid potassium chlorate (KClO3) was heated in a test tube. • 2KClO3(s) → 2KCl (s) + 3O2(g) • The oxygen produced was collected by displacement of water at 22oC at a total pressure of 754 torr. The volume of gas collected was 0.650 L, and the vapor pressure of water at 22oC is 21 torr. Calculate the partial pressure of O2 in the gas collected and the mass of KClO3 that decomposed.
Kinetic Molecular Theory • KMT is a model that helps explain the observed behavior of ideal gases. • It is a simple model which gives us a working approximation of gas behavior. • The particles are so small we can ignore their volume. • The particles are in constant motion and their collisions cause pressure.
Kinetic Molecular Theory • The particles do not affect each other, neither attracting or repelling. • The average kinetic energy is proportional to the Kelvin temperature. • Appendix 2 shows the derivation of the ideal gas law and the definition of temperature.
KMT and: • Pressure and Volume (Boyle’s Law) • Volume and Temperature (Charles’s Law) • Number of moles and volume (Avogadro’s Law)
The Meaning of Temperature • Temperature indicates the average kinetic energy (KE) of the molecules in a gas. • Since KE = 1/2 mv2 and the mass of molecules doesn’t change, an increase in KE means molecules move faster • (KE)avg = 3/2 RT
What it tells us • This the meaning of temperature. • u is the particle velocity. • u is the average particle velocity. • u 2 is the average of the squares of the particle velocities. • the root mean square velocity is • Ö u2 = urms
Range of velocities • The average distance a molecule travels before colliding with another is called the mean free path and is small (near 10-7) • Temperature is an average. There are molecules of many speeds in the average. • Shown on a graph called a velocity distribution
Combine these two equations • (KE)avg = NA(1/2 mu 2 ) • (KE)avg = 3/2 RT Where M is the molar mass in kg/mole, and R has the units 8.3145 J/Kmol. • The velocity will be in m/s
Example • Calculate the root mean square velocity of carbon dioxide at 25ºC. • Calculate the root mean square velocity of hydrogen at 25ºC. • Calculate the root mean square velocity of chlorine at 25ºC.
273 K number of particles Molecular Velocity
273 K 1273 K number of particles Molecular Velocity
273 K 1273 K number of particles 2273 K Molecular Velocity
Velocity • Average increases as temperature increases. • Spread increases as temperature increases.
Effusion • Passage of gas through a small hole, into a vacuum. • The effusion rate measures how fast this happens. • Graham’s Law the rate of effusion is inversely proportional to the square root of the mass of its particles.
Diffusion • The spreading of a gas through a room. • Slow considering molecules move at 100’s of meters per second. • Collisions with other molecules slow down diffusions. • Best estimate of rate of diffusion is Graham’s Law.
Examples • Calculate the ratio of the effusion rates of hydrogen gas (H2) and uranium hexafluoride (UF6), a gas used in the enrichment process to produce fuel for nuclear reactors. • If 0.00251 mol of NH3 effuse through a hole in 2.47 min, how much HCl would effuse in the same time?
Real Gases • Real molecules do take up space and they do interact with each other (especially polar molecules). • Need to add correction factors to the ideal gas law to account for these.