Experiment 19:
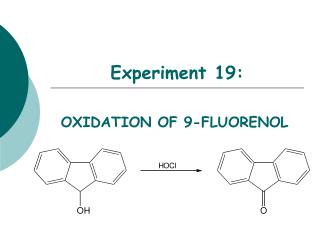
Experiment 19:. OXIDATION OF 9-FLUORENOL. Objectives:. To synthesize a ketone from a secondary alcohol using household bleach. To monitor the reaction progress using TLC analysis. To purify product using simple extraction.

Experiment 19:
E N D
Presentation Transcript
Experiment 19: OXIDATION OF 9-FLUORENOL
Objectives: • To synthesize a ketone from a secondary alcohol using household bleach. • To monitor the reaction progress using TLC analysis. • To purify product using simple extraction. • To analyzethe purity of the product using TLC and melting point analysis. • To characterize reactants and products using IR analysis.
Before coming to lab… • Review these techniques: • TLC analysis • Acid-base Extraction • Drying over MgSO4 • Melting Point Analysis
TYPICAL OXIDATION VS. GREEN OXIDATION Typical oxidation using chromium compound Greener oxidation using household bleach
THE OXIDIZING AGENT NaOCl + CH3CO2H HOCl + CH3CO2Na Sodium acetic hypochlorous sodium Hypochlorite acid acid acetate • Sodium hypochlorite is the main ingredient in bleach. • It must first be converted to hypochlorous acid in order to oxidize the alcohol. • HOCl is a source of a positive Cl, which has 2 fewer electrons than a chloride anion. • Remember, oxidation is the loss of H or the addition of O.
PROPOSED MECHANISM 1 3. Water abstracts a proton from the oxygen bearing the chlorine… 1. Bleach is reacted with acetic acid to produce hypochlorous acid. 2 3 2. Electropositive chlorine atom undergoes nucleophilic attack by oxygen of alcohol to form O-Cl bond… 4. …followed by another attack on the proton of the carbon bearing the oxygen. 4 5. Both electrons from the C-H bond form the p bond of the carbonyl, and a chlorine atom is eliminated to form the neutral product ketone. 5 Although complex, the mechanism results in the exchange of a Cl+ with a H+ on oxygen, followed by subsequent elimination of HCl to form the ketone.
Reason for difference in Rf values The 9-fluorenol can DONATE hydrogen bonds to the silica gel on the TLC plate, resulting in a lower TLC Rf value! The 9-fluorenone can ACCEPT hydrogen bonds, but not donate to the silica gel on the TLC plate, resulting in a higher TLC Rf value!
Reason for difference in color More conjugated Less conjugated • The more conjugated a compound is, the higher the wavelength of light it absorbs. • The visible region of the spectrum is the 400-700 nm wavelength range. • If a compound absorbs light close to 400 nm, it will appear as a yellow color.
EXPERIMENTAL PROCEDURE(SYNTHESIS) • Add Fluorenol to 50 mL flask with stir bar. • Add acetone and stir of until dissolved. • Add acetic acid and bleach while stirring. Place a small cork in top of flask to reduce decomposition of bleach. • Stir 10 minutes. • Perform TLC experiment to check for completion. • If oxidation is incomplete, add more acetic acid and bleach, react for 10 additional minutes and repeat TLC experiment. • If oxidation is complete, proceed to purification steps. Complete reaction!!! RCT STD YOUR RXN PROD STD
EXPERIMENTAL PROCEDURE(PURIFICATION) • Transfer liquid to separatory funnel. • Extract product into diethyl ether 2X. • Wash organic layer with 5% NaHCO3 and Saturated NaCl. • Transfer organic layer to clean flask. • Dry over MgSO4. • Transfer liquid to preweighed 150 mL beaker w/3 boiling chips. 125 mL 150 mL
EXPERIMENTAL PROCEDURE(PURIFICATION) • Prepare a tap water bath using your 400mL beaker. • Place the water bath on a hotplate and heat the water on a setting of 3 (NO HIGHER) until just above room temp. • Remove water bath from hotplate. • Slowly place the 150mL beaker containing diethyl ether into the water bath to evaporate the liquid solvent. • If the solution boils too rapidly, remove it immediately until it ceases to boil! 150 mL 400 mL
EXPERIMENTAL PROCEDURE(PRODUCT ANALYSIS) • Place LABELED 150mL beaker in oven for 10 minutes to dry. Reweigh. • Repeat if %yield is greater than 100%! • Reweigh to obtain final product mass and calculate % yield. • Perform melting point analysis. YOUR NAME HERE
IR Spectroscopy • Remember to concentrate on the types of bonds that indicate the CONVERSION from reactant to product! 3041 3196 1031 3060 1716
SAFETY CONCERNS • DIETHYL ETHER IS EXTREMELY FLAMMABLE! NEVER place diethyl ether directly on a hotplate! • Acetic acid is corrosive!
WASTE MANAGEMENT • “LIQUID WASTE” container: any/all liquid waste from extraction and TLC experiment. • “SOLID WASTE” container: any/all solid waste from synthesis and purification. • BROKEN GLASS box: Place used TLC and melting point capillaries.
CLEANING • Clean all glassware with soap, water, and brush if necessary. • Rinse all glassware with wash acetone before returning to lab drawer. • DO NOT return any glassware to lab drawer dirty or wet.